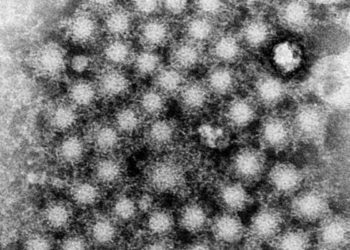
Adenovirus encoding hepatitis C virus vaccine is safe but effectiveness is unclear
1. In this phase 1-2 clinical trial, an adenovirus viral vector expressing non-structural hepatitis C proteins was safe in a high-risk population.
2. The vaccine did not have a significant impact on chronic hepatitis C infection prevention.
Evidence Rating Level: 1 (Excellent)
Study Rundown: The hepatitis C virus (HCV) is one of the most prevalent blood-borne infections while conferring significant mortality and morbidity. A vaccine strategy is important in reducing the spread among high-risk populations and may be essential in the goal of eliminating the disease. In this randomized trial, participants were either assigned to a recombinant chimpanzee adenovirus 3 vector or placebo group. At six months, there was no significant difference in the rates of chronic HCV infection between vaccine and placebo groups. T-cell responses were detected in the vaccine group and a minimal amount was seen in the placebo group. The percentages of participants with serious adverse events were similar between both groups. The most common serious adverse event was related to increased alanine transaminase (ALT) levels. The limitation of the study was the inability to reveal some level of efficacy in the prevention of HCV infections even though the strategy was shown to be safe. Overall, this trial demonstrates that this vaccine strategy has no evidence of efficacy and highlights the difficulty in formulating an HCV vaccine given chronic infection involves likely immune-escape mechanisms.
Click here to read the study in the NEJM
Relevant Reading: Subinfectious hepatitis C virus exposures suppress T cell responses against subsequent acute infection
In-Depth [randomized controlled trial]: In this phase 1-2, double-blinded, placebo-controlled randomized trial of 548 participants, participants at risk of HCV infection were enrolled based on a history of recent intravenous drug use were assigned 1:1 to receive vaccine or placebo in a day 0 and day 56 regiment. The vaccine was a recombinant chimpanzee adenovirus 3 vector followed by a recombinant modified vaccinia Ankara boost encoding non-structural proteins of the HCV. The primary endpoint was the rate of chronic HCV infection at 6 months defined by HCV RNA levels. The rate of chronic HCV infection was similar between both groups (hazard ratio [HR] 1.53, 95% confidence interval (CI) of 0.66 to 3.55). The geometric mean peak HCV RNA level was 152 x 103 international units (IU) per milliliter (mL) in the vaccine group compared to 1804 x 103 IU per mL in the placebo group. Immunogenicity was assessed before viral infection. T-cell responses were detected in 78% of the vaccine group and only in 3% of the placebo group. The rate of serious adverse events was similar between both groups, with most events related to a laboratory detected adverse event. The most frequent laboratory detected adverse event was an elevation in the ALT levels which was detected in 50% of the vaccine group and 39% in the placebo group. Overall, the trial determined the adenovirus vaccine strategy was safe; however the effectiveness to prevent subsequent HCV infection was not able to be determined.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.