Approved label populations may be broader than trial populations for new drugs
1. In this cross-sectional study, approved label populations were broader overall compared to trial populations for new drugs in the United States and Europe.
2. The category of patient fitness most often exhibited broader label populations than trial populations, followed by disease severity.
Evidence Rating Level: 3 (Average)
Study Rundown: The United States (US) Food and Drug Administration (FDA), the European Medicines Agency (EMA), and Switzerland’s Swissmedic are three major drug regulatory agencies which appraise and approve new drugs based on clinical trial data regarding their quality, safety, and efficacy. When approving a new medication, a broader indication, or the “label indication,” may be extrapolated and approved from the narrower trial population with respect to various criteria including disease subtype and severity. Extrapolation can be justified in circumstances where the clinical evidence suggests that trial findings can be applied to populations excluded from the study or in cases where it is difficult to include all patient subpopulations due to the rarity of the disease. However, populations excluded from the study may not experience the expected benefits or be subject to increased risk of adverse events compared to the trial populations. Prior studies investigating the prevalence of label indications have been limited to drugs approved by the FDA for a short time period or specific drug classes, and the role of extrapolation in drug approval at the EMA or Swissmedic remains unclear. Hence, this cross-sectional study aimed to analyze the differences in the characteristics of enrolled patients including age, disease subtype, disease severity, prior therapy and patient fitness between trial populations and approved label populations in the US, European Union (EU), and Switzerland for new drugs. Overall, it demonstrated that approved label populations were broader compared to trial populations for new drugs, especially in the US compared to the EU and Switzerland. The category of patient fitness most often had greater variance than trial populations, followed by disease severity. The study was limited by a lack of uniformity in the reporting of trial eligibility criteria. Overall, these results highlighted the need for continued post-approval surveillance of medications to ensure safety and effectiveness.
Click to read the study in AIM
Relevant Reading: Use of extrapolation in new drug approvals by the US Food and Drug Administration
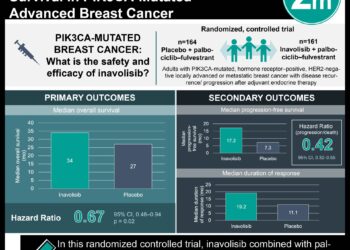
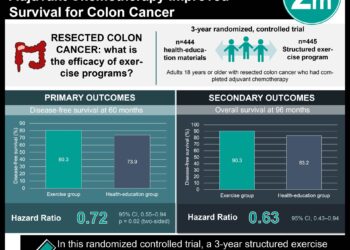
In-Depth [cross-sectional study]: This cross-sectional study analyzed differences in enrolled patient characteristics between trial and approved label populations in the US, EU, and Switzerland for new drugs. Among all new drugs with novel active ingredients approved by the FDA between January 1, 2012 and December 31, 2023, those which were also approved by the EMA and Swissmedic during the same period were included. The eligibility criteria from the efficacy trials were classified into five categories including age, disease subtype, disease severity, prior therapy, and patient fitness. An indication was appraised as “broader” if the label population was more extensive in at least one category compared to the eligibility criteria for the trial population, “more restrictive” if the label population was more restrictive in at least one category compared the eligibility criteria for the trial population, and “congruent” if the label and trial populations matched. A score of +1 was assigned for broader, -1 for more restrictive, and 0 for congruent for each category and a cumulative score for the five categories was obtained. Of 520 drugs approved by the FDA during the inclusion period, 263 were also approved by the EMA and Swissmedic for 278 indications. All three agencies had a cumulative score greater than 0, including a composite of 1.83 for the FDA (95% confidence interval [CI], 1.68 to 1.98), 1.39 for the EMA (95% CI, 1.23 to 1.55), and 1.31 for Swissmedic (95% CI, 1.14 to 1.47). Patient fitness most often had broader label populations with 276 approved indications in the US (99.3% [CI, 99.2% to 99.4%]), 276 in the EU (99.3% [CI, 99.2% to 99.4%]), and 275 in Switzerland (98.9% [CI, 98.8% to 99%]), followed by disease severity with 143 approved indications (51% [CI, 49% to 54%]), 119 approved indications (43%, [CI, 40% to 46%]), and 111 approved indications (40% [CI, 37% to 43%]), respectively. In summary, this trial demonstrates that label populations were broader than trial populations for new drugs approved in the US and Europe.
Image: PD
©2025 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.



![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-75x75.png)

