Bivalirudin not linked to adverse cardiovascular events: The MATRIX program
1. Acute coronary syndrome (ACS) patients requiring percutaneous coronary intervention (PCI) who received either heparin or bivalirudin were not significantly different in either the rates of major adverse cardiovascular events and overall adverse clinical events.
2. Use of bivalirudin in patients with ACS requiring PCI yielded a significantly lower rate of any-cause death as well as cardiac death compared to patients receiving heparin.
Evidence Rating Level: 1 (Excellent)
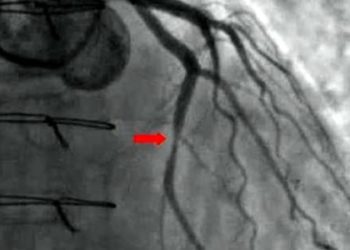
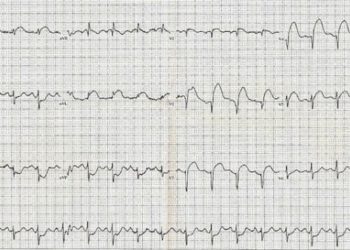
Study Rundown: The use of unfractionated heparin versus bivalirudin with a glycoprotein IIb/IIIa inhibitor are two of the most common antithrombotic regimens used worldwide for patients with acute coronary syndrome. Existing literature has yet to produce a definitive answer as to which approach is superior with regards to reducing ischemic, bleeding or mixed adverse outcomes. This large multicenter study investigated whether bivalirudin is superior to unfractionated heparin and discretionary use of glycoprotein IIb/IIIa inhibitors. This study found no significant differences among patients with ACS, patients undergoing PCI for major adverse cardiovascular events as a composite of any-cause death, MI and stroke, and no difference between net adverse clinical events, when heparin or bivalirudin were used. A statistically significant lower rate of any-cause death and cardiac death was observed in the bivalirudin group compared to the heparin group. The study results are limited by the use of pre-PCI glycoprotein IIb/IIIa inhibitors in the heparin group per physician judgment, as well as the use of two different regiments for post-PCI bivalirudin infusion.
Click to read the study in NEJM
In-Depth [randomized controlled trial]: Minimizing Adverse Hemorrhagic Events by Transradial Access Site and Systemic Implementation of Angiox (MATRIX) was a multicenter, randomized, open-label trial. A total of 7213 patients with ACS requiring PCI were randomly assigned to receive either bivalirudin or unfractionated heparin with PCI. Participants included NSTEMI patients with worsening ischemia and associated risk factors for cardiac disease as well as STEMI patients presenting within a limited timeframe from symptom onset. Follow-up occurred at 30 days and information for only 7188 of the participants were available. Primary outcomes included composite death from any cause or causes secondary to stroke or MI. Additional primary outcomes included adverse clinical events including major bleeding unrelated to CABG. Secondary outcomes included individual components of composites from the primary outcomes. For primary outcomes, there was no significant difference in major adverse cardiovascular events between the bivalirudin versus heparin groups (RR = 0.94; 95% [CI] = 0.81-1.09; p=0.44). Similarly, there was no difference in secondary outcomes for net adverse events between the two groups (RR = 0.89; 95% [CI] = 0.78-1.03; p=0.12). However, the rate of death from any cause was significantly lower in the bivalirudin group (RR=0.71, 95% [CI] = 0.51-0.99, p=0.04) as was the rate of death from cardiac causes (RR= 0.68; 95% [CI] = 0.48-0.97; p=0.03). This lower rate of death was associated with lower rates of bleeding in the bivalirudin group. The trial included two different regiments of post-PCI bivalirudin infusion in the bivalirudin group and for discretionary use of glycoprotein IIb/IIIa inhibitors in the heparin group which were consistent with the standard of practice but made interpretation of the data difficult.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.