Brentuximab vedotin improves progression-free survival in Hodgkin’s lymphoma patients [AETHERA trial]
1. In this phase 3 randomized controlled clinical trial, brentuximab vedotin significantly improved progression-free survival in Hodgkin’s lymphoma patients at risk of relapse/progression after autologous stem-cell transplantation.
2. Notable adverse events associated with this treatment included peripheral sensory neuropathy and neutropenia. Deaths from treatment-related acute respiratory distress syndrome were also noted.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Hodgkin’s lymphoma is a type of cancer affecting lymphocytes with variable prognosis. The current standard of care involves a combination of radiation therapy, chemotherapy (induction and salvage), and hematopoietic stem cell transplantation. In a phase 3 clinical trial published today in Lancet, brentuximab vedotin (BV) – the first new drug for Hodgkin’s lymphoma in 30 years – was shown to significantly prolong progression-free survival in Hodgkin’s lymphoma patients who were at risk of relapse or progression after autologous stem cell transplantation. If ultimately approved, BV adds to an existing arsenal of therapies that continue to improve the prognoses for patients afflicted by Hodgkin’s lymphoma. The study was a well-powered randomized, double-blind, placebo-controlled trial and was intended to establish causality between intervention and outcome by design. Despite the promising results, a possible confounder was a slight variation in randomization – a higher proportion of female patients and black patients were assigned to the BV group. In addition, the study only included patients that had received autologous stem cell transplantations. The benefits for patients following heterologous transplants remain unclear. Finally, Hodgkin’s lymphoma is a “basket” diagnosis based on histological features and cell surface markers, and contains at least 5 distinct disease entities per WHO classification. As a result, further studies are needed to identify patients that are most likely to benefit from this new agent based on subtypes.
The study was funded by Seattle genetics and Takeda Pharmaceuticals International. The funders had a role in study design, data analysis, data interpretation, and writing of the report.
Click to read the study in the Lancet
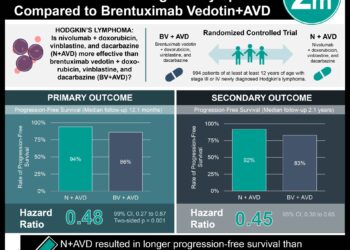
In-Depth [randomized controlled trial]: This study enrolled 329 patients with histology-confirmed Hodgkin’s lymphoma who had previously undergone high-dose therapy followed by autologous hematopoietic stem-cell transplantations. Patients were randomized 1:1 to receive either 1.8 mg/kg IV brentuximab vedotin (an anti-CD30 antibody) or placebo. Patients were then reassessed at baseline and at months 3, 6, 9, 12, 18, and 24 months after first dose for progression. The primary endpoint was progression-free survival.
The primary endpoint was significantly improved in patients receiving BV (stratified HR 0.57, 95%CI 0.40-0.81, p = 0.0013). The median progression-free survival in the BV group was 42.9 months (95%CI, 30.4-42.9), compared to 24.1 months (95%CI, 11.5-not estimable) for placebo. The estimated 2-year rate of progression-free survival was 63% (95%CI 55-70%) for the BV group and 51% (95%CI 43-59%) for the placebo group.
In safety analyses, the most common adverse events in the BV group was peripheral sensory neuropathy (112 or 67% of patients in the BV group vs. 31 or 19% of the placebo group). Additionally, neutropenia was more common in the BV group, with grade 3 or higher neutropenia in 49 (29%) patients in the BV group, compared to 16 (10%) in the placebo group. There were 2 deaths resulting from treatment-related acute respiratory distress syndrome.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.



![Hypertension drug guanabenz may improve multiple sclerosis outcomes [Pre Clinical]](https://www.2minutemedicine.com/wp-content/uploads/2015/03/neuron3_edited-75x75.jpg)
