Camrelizumab with chemotherapy demonstrated improved overall survival and progression-free survival of esophageal squamous cell carcinoma patients versus placebo
1. Camrelizumab improved the overall survival and the progression-free survival of esophageal squamous cell carcinoma patients compared to placebo treatment as adjuvants to chemotherapy.
2. Grade 3 adverse events were comparably common in the camrelizumab treated group as the placebo group, but there was a greater proportion of pneumonitis, a serious adverse event, in the camrelizumab group.
Evidence Rating Level: 1 (Excellent)
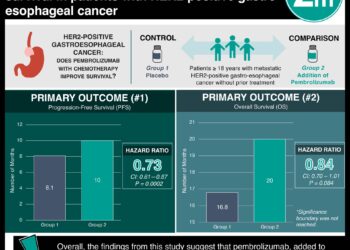
Study Rundown: Esophageal squamous cell carcinoma (ESCC) is the most common type of esophagus cancer in Asia, which in turn is a common cause of death. Survival on the current standard of care, 2 cytotoxic chemotherapy agents, is 7.0-13.0 months. Camrelizumab is a selective IgG4-κ monoclonal antibody against programmed death receptor 1, which is used to treat other cancers and has been approved to treat ESCC in China. This study investigated the efficacy (including progression free survival (PFS) and overall survival (OS)) and adverse events of a combination of camrelizumab and chemotherapy to treat ESCC. Half of 594 adults with confirmed ESCC and sufficient life-expectancy were randomized to receive camrelizumab, while the remainder received placebo, with their chemotherapy. Tumor progression was imaged to assess for PFS for a median of 10.8 months. Camrelizumab improved OS and PFS compared to placebo with OS at 18 months of 42.7% and 29.5% and PFS at study end of 6.9 months and 5.6 months, respectively. 47.3% of the camrelizumab patients compared to 63.4% of the placebo group had to discontinue their allotted treatment due to disease progression. 99.3% of the camrelizumab group experienced adverse events, compared to 97.0% of the placebo group, with the most common being decreased neutrophil count, white blood cell count, anemia, and rarely pneumonitis. Camrelizumab improved global health status, pain, and choking in ESCC patients compared to the placebo. A limitation of this study is that a non-differential set of patients received checkpoint inhibitor treatment after camrelizumab or placebo treatment that could improve the overall survival scores. Additionally, other medical management such as in-/out-patient status, activity, and socio-economic status were not considered, even in subgroup analysis. Finally, the scope of this study is limited to those with ESCC without other conditions and a minimum of a 12-week life expectancy, so it does not report on how this treatment would affect the most advanced cases of ESCC.
Click to read the study in JAMA
Click to read an accompanying editorial in JAMA
In-Depth [randomized controlled trial]: This double-blind, multi-site, phase 3 clinical trial was conducted in China. Eligible participants included those aged 18-75 with histologically or cytologically confirmed ESCC. Patients with other malignancies, autoimmune disease, central nervous system metastasis, organ failure, or a life expectancy of <12 weeks and were excluded. 298 were randomized to a 200 mg camrelizumab-chemotherapy (175 mg/m2 IV paclitaxel and 75 mg/m2 IV cisplatin for 6 of 3-week cycles) group and 298 participants were randomized to a placebo-chemotherapy group. Radiography was used to assess tumor progression every 6 weeks. Patients in the camrelizumab and placebo group had comparable baseline demographics and disease characteristics. The study followed patients for a median of 10.8 months. 26.2% of the camrelizumab patients and 9.1% of the placebo group continued their treatment. 47.3% of the camrelizumab patients and 63.4% of the placebo group discontinued their treatment due to disease progression. OS was improved by camrelizumab treatment compared to placebo (45.3% vs 58.4% death rate; OS = 15.3 months, 95% CI = 12.8-17.3 months vs OS = 12.0 months, 95% CI = 11.0-13.3 months; HR = 0.70, 95% CI = 0.56-0.88; P = 0.001). The probability of OS at 18 months was 42.7% (95% CI = 35.3%-50.0%) in the camrelizumab group compared to 29.5% (95% CI = 22.9%-36.3%) in the placebo group. The PFS was improved by camrelizumab treatment compared to placebo (66.8% vs 76.8% progression or death rate; PFS = 6.9 months, 95% CI = 5.8-7.4 vs PFS = 5.6 months, 95% CI = 5.5-5.7; HR = 0.56, 95% CI = 0.46-0.68; P < 0.001). Objective response to treatment occurred in 72.1% of the camrelizumab group (95% CI = 66.7%-77.2%) and a comparable 62.1% of the placebo group (95% CI = 56.3%-67.6%; camrelizumab vs placebo: 95% CI = 2.6%-17.6%, P = 0.009). The disease control rate was similar between the camrelizumab and placebo groups (91.3%, 95% CI = 87.5%-94.2% vs 88.9%, 95% CI = 84.8%-92.3%). Adverse events occurred in 99.3% of the camrelizumab group compared to 97.0% of the placebo group. The most common Grade 3 adverse events were decreased neutrophil count (39.9% vs 43.4%), decreased white blood cell count (24.2% vs 26.6%), and anemia (17.4% vs 13.5%), while the most common serious adverse event was pneumonitis (5.7% vs 2.7%). Adverse events interrupted treatment in 45.3% of the camrelizumab group and 23.9% of the placebo group and lead to death in 3.0% and 3.7% of the groups, respectively. On quality-of-life surveys, there was a 2.6 point improvement in the global health status (95% CI = 0.0-5.2), a 3.1 point reduction in pain (95% CI = −5.3-−0.9), and a 3.4 point reduction in reported choking while eating/swallowing (95% CI = −5.9-−0.8).
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.