Low success and satisfaction with intrauterine ball contraception
1. The experimental intrauterine ball had high expulsion rates, high rates of removal due to symptoms, and low satisfaction rates during the first year of use.
Evidence Rating Level: 4 (Below Average)
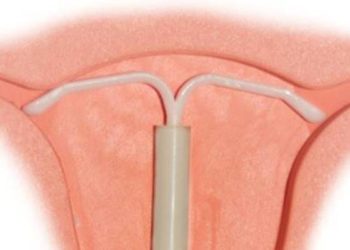
Study Rundown: The American College of Obstetrics and Gynecologists (ACOG) recommends long acting reversible contraceptive methods (LARCs) as first line birth control for women of all ages. They are over 99 percent effective and very well tolerated by most women. Current LARC methods available in the United States include a hormonal implant (Nexplanon), which is inserted under the skin of the inner arm, a copper intrauterine device (Paragard IUD), and two hormonal IUDs (Mirena and Skyla). While IUDs offer numerous benefits, they aren’t without downsides. Common complications include uterine perforation, infection, and expulsion. Additionally, the copper IUD can cause side effects like increased bleeding and cramping–up to 20 percent of women discontinue the copper IUD within the first year because of such complaints. The intrauterine ball (IUB) is a novel, non-hormonal contraception device that was designed to function similar to a copper IUD, but eliminate some of the complications and side effects that lead to discontinuation. In theory, its three-dimensional spherical shape and elasticity allow it to conform to the uterus and reduce expulsion and tissue irritation, while the insertion device curves down, away from the uterine fundus, as to reduce perforations. In this pilot study, researchers sought to test the SCu380A, a 12 mm diameter IUB, for method-related discontinuation rates, in preparation for a larger efficacy trial.
In this pilot study, researchers found that the IUB had high expulsion rates, high rates of removal due to symptoms, and low satisfaction rates during the first year. Authors concluded that these findings suggest the IUB is an unacceptable alternative to current intrauterine contraceptive methods. Strengths included the novel contraceptive design—innovations have the potential to improve LARC methods and increase uptake. Limitations included the high (14%) loss to follow-up rate, even accounting for these women the expulsion rates and discontinuation rates remained high. Future studies testing modifications of the design are currently underway.
Click to read the study in Contraception
Relevant Reading: The IUB, a newly invented IUD
In-Depth [prospective pilot study]: Fifty-one women had a 12 mm diameter experimental intrauterine ball contraceptive device (SCu380A) inserted in their uterus by a single provider. Ultrasound post insertion confirmed correct placement. Follow-up was scheduled at 6-8 weeks, 3, 6, 9 and 12 months. The primary outcome was method-related discontinuation. The secondary outcome was patient satisfaction.
At 8 weeks there were 9 expulsions (18%) and 2 removed for symptoms of pain and bleeding (4%). At 12 months there were 14 expulsions (28%), and 8 removals for symptoms (16%). Only 21 women retained their IUB at 12 months (48%), 15 of whom were satisfied with the method (71%), Complaints of those who were not satisfied included bleeding, spotting, and pain.
Image: CC/flikr/TipsTimesAdmin
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.