Non-TNF-targeted therapies more effective than anti-TNF agents in uncontrolled rheumatoid arthritis
1. The addition of a non-TNF-targeted biologic therapy is superior to the addition of an anti-TNF biologic therapy in patients with rheumatoid arthritis that is not well controlled with a first-line anti-TNF medication.
2. There was no difference in serious adverse events or infections between the two classes of therapeutics.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Patients with poorly controlled rheumatoid arthritis (RA) pose a significant challenge for the medical community. This study sought to determine whether patients with RA that is not controlled with a first-line anti-TNF therapy would benefit more from the addition of another anti-TNF therapy (adalimumab, etanercept, infliximab, etc.) or by a non-TNF-targeted therapy (abatacept, rituximab, etc.). While the specific biologic therapy in each class was up to the discretion of treating physicians, this study found that patients who received a non-TNF-targeted therapy had higher rates of response and lower overall levels of ongoing disease activity.
This study shows a clear benefit of non-TNF-targeted therapies for patients with poorly controlled RA. Its results are strengthened by a rigorous randomization protocol and follow-up of disease progression over 52 weeks. However, the study was conducted in a non-blinded fashion which could potentially add to physician and patient bias, especially given the subjective nature of the efficacy. Further, large-scale randomized controlled trials are needed to further evaluate the validity of these findings.
Click to read the study in JAMA
Relevant Reading: Rheumatoid Arthritis
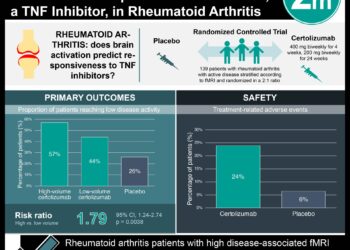
In-Depth [randomized controlled trial]: This study was an open-label randomized clinical trial that enrolled 300 patients with rheumatoid arthritis that was not controlled on a TNF-alpha inhibitor. These patients were randomized to receive an additional anti-TNF drug or an additional non-TNF-targeted drug. Patients were followed for a total of 52 weeks. The primary outcome was the proportion of patients who achieved good or moderate response to therapy at 24 weeks. Therapy response was determined by subjective patient ratings on the European League Against Rheumatism (EULAR) scale. Secondary outcomes consisted of EULAR response at 12 and 52 weeks, achievement of low disease activity (DAS28 <3.2) or remission (DAS28 <2.6), serious adverse events, and serious infections.
The results demonstrated that patients receiving a non-TNF-targeted biologic had higher rates of good or moderate EULAR response at 24 weeks (69% vs. 52%, OR 2.06; p = 0.004). This difference in response remained significant at 52 weeks, when 41% of patients in the non-TNF-targeted biologic cohort reported low disease activity compared to only 23% in the anti-TNF therapy arm (OR 2.26, p = 0.003). There was no statistical difference in adverse events or serious infections between the two groups.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.