Novel hepatitis C treatment may be cost-effective in early stages of fibrosis
1. Based on modeling data from the treatment trials of HCV genotype 1 infection with the newer agents, treating even at the early stages of fibrosis is cost-effective (spending <$50,000 per quality-adjusted life year was considered cost-effective) compared to waiting for late stages of fibrosis.
2. Treating about 50% of the eligible patient population at any stage of fibrosis would cost about $50 billion, compared to about $30 billion if the 50% of advanced fibrosis stage patients were treated.
Evidence Rating Level: 2 (Good)
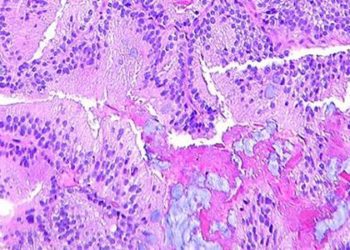
Study Rundown: Hepatitis C (HCV) infection is the leading cause of liver-related deaths, hepatocellular carcinoma, and liver transplants in the US. The latest therapies, which include HCV nucleotide analogue nonstructural protein 5A and B inhibitors, lead to a greater than 90% cure rate for HCV genotype 1, but cost about $1000 per day or more. While there are studies that show these newer drugs can be cost-effective, the optimal time for treatment, in regards to level of fibrosis (F0 = no fibrosis, F4 = cirrhosis) is not known. This analysis, which modeled treatment of HCV at all stages of liver fibrosis compared to advanced stages, was conducted to help determine the optimal level of liver fibrosis at which to introduce HCV therapies from a cost-benefit perspective. The results suggested that, even at the early stages of fibrosis, treating HCV infection with the newer drugs was cost-effective in the long run (spending <$50,000 per quality-adjusted life year was considered cost-effective). Also, treating about 50% of the eligible patient population at any stage of fibrosis would cost about $50 billion, compared to about $30 billion if the 50% of advanced fibrosis stage patients were treated.
The model had several limitations. There was limited data on the long-term efficacy of an initial sustained-virologic response, which may overestimate the savings. Additionally, the models were based off of meta-analyses of studies, which may be “best-case scenarios” and may not accurately reflect real life scenarios. Additionally, the costs of the drugs may be different based on health network negotiations, and may change over a few years, which would make the results difficult to interpret.
Click to read the study in JAMA Internal Medicine
Relevant Reading: Cost-effectiveness and budget impact of hepatitis C virus treatment with sofosbuvir and ledipasvir in the United States
In-Depth [decision-analytic model]: Investigators modeled the approved newer treatments for HCV genotype 1 amongst treatment-naïve Americans at different levels of fibrosis. The fibrosis levels were defined as follows: F0 = no fibrosis, F1 = portal fibrosis without septa, F2 = portal fibrosis with rare septa, F3 = numerous septa without cirrhosis, F4 = cirrhosis. The likelihood of sustained virologic response (SVR) and discontinuation of therapy were based off of meta-analyses of the clinical trials that initially evaluated this therapy.
Specifically, the model evaluated sofosbuvir-ledipasvir treatment for 8 or 12 weeks (depending on viral load). The modeled natural progression of disease and mortality were also based off the published literature. The model outcomes were quality-adjusted life years (QALYs) and health care cost. Treating any stage of fibrosis compared to just F3 and F4 produced a QALY gain of 0.73. The cost, however, of treating all fibrosis stages lead to a cost increase of $39,475 per QALY gained.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.