Radiographic surrogates may predict tumor-specific survival in renal carcinoma
1. Computed tomography derived imaging characteristics were correlated with disease specific gene expression to create a radiogenomic based surrogate of molecular assays (SOMA) in patients with clear-cell renal cell carcinoma (CCRCC) which was predictive of disease-specific and stage-independent survival in a validated cohort.
2. Radiogenomic based SOMAs can theoretically be created for different disease entities without the need for invasive tissue acquisition, providing new clinical tools for disease stratification and individual prognostication in clinical trials.
Evidence Rating: 2 (Good)
Study Rundown: Genomic tumor information has increasingly been used to help in the prognostication, stratification, and delineation of treatment strategies for patients, but has been limited by invasive tissue acquisition and by the lack of macrophenotypic data often revealed by imaging. To circumvent these difficulties and integrate the value of non-invasive imaging, SOMAs can be developed based on correlation of specific imaging characteristics to individual genomic indicators within a cohort. This allows for a non-invasive, quantitative tool to integrate the patient-specific information offered by both genomic and radiologic data to help in the prognostication and stratification of specific tumors. In this study, a SOMA was designed for patients with CCRCC based on a known cohort of 70 patients with surgically resected CCRCC who underwent pre-operative CT imaging and molecular characterization of surgical pathology samples. A radiogenomic risk score (RRS) model that predicted CCRCC-specific survival in a stage-, grade- and performance status independent fashion was developed from the known CCRCC cohort, and then validated in a prospective CCRCC cohort. This study, while suggesting immediately relevant prognostication tools for CCRCC, also suggests that similar radiogenomic surrogates can be created for other tumors across modalities, helping create prognostication tools that do not necessarily rely on tissue acquisition for each patient. This study was limited by its overall size, and by its population selection, which may limit the generalizability of this RRS for patients with CCRCC from other populations. Future studies should validate the approach with a more heterogeneous multi-center population and determine if the RRS can be extrapolated to larger populations, or should be reserved for geographically limited populations.
Click to read the study in Radiology
Relevant reading: Molecular stratification of clear cell renal cell carcinoma by consensus clustering reveals distinct subtypes and survival patterns
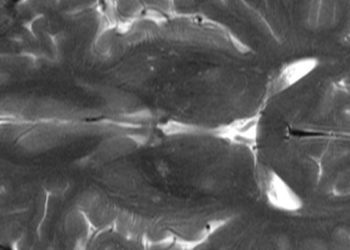
In-Depth [retrospective cohort]: Based on analysis and regression of gene expression analysis and CT imaging trait analysis, an RRS model was developed, with four key imaging features that were independent of stage, grade, and patient performance status, specifically: quartile percentage of tumor necrosis, tumor transition zone, tumor-parenchyma interaction, and tumor-parenchyma interface. Briefly, each of these features were chosen to represent physiologic and morphologic characteristics of the local tumor microenvironment and the tumor itself, and were correlated by multivariate linear regression to the top eigenvector of each patient’s individual gene expression matrix. The gene expression matrix was independently correlated to patient outcomes in a separate publication. In short, this mathematically linked the expression of specific risk-associated genes with specific radiophenotypic features identified by CT imaging to generate the RRS. This risk score was then validated in a prospective cohort of patients with CCRCC, showing that there was a relationship between RRS and a previously and validated genomic-based risk score (R = 0.45, p < 0.001, classification accuracy of 68.6%, p < 0.001), and that the RRS could predict disease-specific survival (log-rank p < 0.001), and was independent of stage, grade, and performance status (multivariate Cox model p < 0.05, log-rank p < 0.001).
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.