Single course intravenous immunoglobulin ineffective at controlling severe solar urticaria
1. Out of 9 patients with severe solar urticaria (SU) who received a single-dose of intravenous immunoglobulin (IVIG), 2 showed remission with photo-testing at 12 weeks, with only one patient who showed sustained remission at 48 weeks.
2. Although most patients did not achieve full remission at 4 weeks, over half the cohort did report improvement in quality of life.
Evidence Rating Level: 2 (Good)
Study Rundown: Solar urticaria (SU) is an uncommon disorder in which wheals develop shortly after exposure to visible light or UV radiation. In patients who have severe disease that is refractory to treatment with antihistamines or other conservative measures, it has been reported that IVIG may be of utility. However, these findings were demonstrated mainly in retrospective case series with limited patients. These authors aimed to study the efficacy of a single dose of IVIG in patients with solar urticaria, specifically investigating the ability of IVIG to induce remission of SU measured by phototesting. The results showed minimal rate of full remission with this method, however about half of the patients did see some clinical response. The study is limited by its small patient pool and lack of a control arm. Future studies are needed to assess rate of response and remission of SU with multiple doses of IVIG.
Click to read the study in Journal of the American Academy of Dermatology
Relevant Reading: Solar urticaria treated with intravenous immunoglobulins
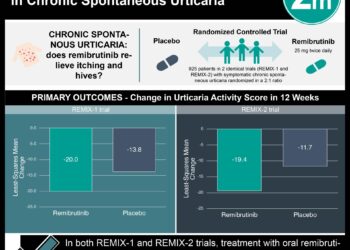
In-Depth [prospective cohort]: 9 patients, mean age 39.4, with severe SU, received a single dose of IVIG (total dose 2 g/kg). Patients were subsequently evaluated by the following metrics: minimal urticarial dose (MUD) calculated by phototesting, Dermatology Life Quality Index (DLQI), disease activity, and intensity of SU by visual analog scale (VAS) at 4 and 12 weeks. 22.2% of the cohort had remission of SU on phototesting at 4 and 12 weeks (95% CI: 2.8-60.0%). 44.4% of patients reported clinical improvement in severity of SU at 4 weeks, with a subsequent drop to 22.2% at 12 weeks. 55.6% of patients similarly reported DLQI of <6 at 4 weeks, as compared to the mean baseline DLQI that was 14.2 (range: 9-23). Notably, about half patients experienced severe headache after treatment with 1 patient who reported aseptic meningitis.
Image: PD
More from this author: Tech-aided surveillance of patients at high risk for melanoma aids early diagnosis, Indoor tanning linked to high skin cancer risk in US and abroad, Free drug samples may alter prescription habits of dermatologists, Atopic dermatitis may be more persistent than previously understood
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.


![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-350x250.png)