2 Minute Medicine Rewind June 22 – June 28, 2015
PD-1 Blockade in Tumors with Mismatch-Repair Deficiency
Cancer immunotherapy seeks to identify unique antigens on malignant cells and disinhibit patient’s immune system responses to unique antigens. It is postulated that highly immunogenic cancers, such as cancers with a large number of somatic mutations due to mismatch repair defects, are more readily targeted by the immune system when disinhibiting immune checkpoints. In this multi-center phase II trial of colorectal cancers with and without mismatch repair deficiencies as well as other cancers with mismatch repair deficiencies, 41 patients with metastatic disease were administered pembrolizumab, an anti-programmed death 1 immune checkpoint inhibitor. There was a marked difference in progression free survival (PFS) and overall survival (OS) in patients with mismatch-repair defects (median PFS and OS not reached) compared to patients without mismatch-repair defects (2.2months and 5.0months, HR 0.1 (p < 0.001) and 0.22 (p = 0.05) respectively). Patients with a mismatch-repair defects, regardless of colorectal origin or otherwise, had a higher mutation burden than patients without mismatch-repair defects (1782 vs. 73 somatic mutations, p = 0.007). The presence of mismatch repair deficiencies and somatic mutation predicts response to pembrolizumab.
Implantable cardioverter-defibrillators (ICDs) are indicated in patients with congestive heart failure with low ejection fractions after optimal medical therapy. Ischemic cardiomyopathy the most common etiology of congestive heart failure, however during the acute post-ACS period, patients are yet to be medically optimized and ICD implantation may be neglected. In this retrospective analysis of national Medicare beneficiaries between 2007 and 2010, investigators identified 10,318 patients with an ejection fraction of 35% or less after myocardial infarction. Only 8.1% of patients received an ICD implantation within a year of myocardial infarction, and were more likely to have had prior coronary artery bypass surgery, higher peak troponin levels, cardiogenic shock, and close post-discharge cardiology follow-up. Patients with ICD placement had lower 2-year mortality (15.3 events per 100 patient-years vs. 26.4 events per 100 patient-years, adjusted HR 0.64, 95% CI 0.53-0.78). Investigators suggest further research should be done to understand how ICD placement can be increased to decrease mortality.
The Strength of Association Between Surrogate End Points and Survival in Oncology
Surrogate end points are often questioned in clinical trials as, despite the best intentions, there are historical examples of improving surrogate endpoints without benefit to clinical outcomes such as mortality. Despite these concerns, surrogate end points in clinical trials are increasingly becoming common in oncology clinical trials. In this meta-analysis of 684 randomized clinical trials, investigators identified 352 studies with both surrogate end points and overall survival data and calculated correlations. Only 23 percent (15 of 65) of surrogate endpoints were highly correlated with survival (r > 0.85), while more than half (52%, 34 of 65) of reported correlations were weakly correlated with survival (r < 0.7). Looking at randomized clinical trial data, there is limited evidence supporting surrogate clinical markers as they are poorly correlated with survival.
High dose methylprednisolone is used to treat multiple sclerosis relapses, and currently is performed in an inpatient setting with intravenous formulations of methylprednisolone. In this multicenter, randomized control trial of adult patients with multiple sclerosis relapse, 199 study participants were randomized to 1g of either oral or intravenous methylprednisolone and were followed for evidence of clinical improvement and the need for repeat steroid use. Study participants, on average, underwent treatment 7.2 days from relapse. There was no statistically significant difference in the proportion of patients who had clinical improvement on the Kurtzke Functional System Scale at 28 days (81% in oral methylprednisolone group vs. 80% in intravenous methylprednisolone group). The investigators found that the efficacy and safety profile of oral vs. intravenous methylprednisolone for patients with multiple sclerosis relapse was similar but potentially could be more cost effective.
Mass Screening for Untreated Atrial Fibrillation The STROKESTOP Study
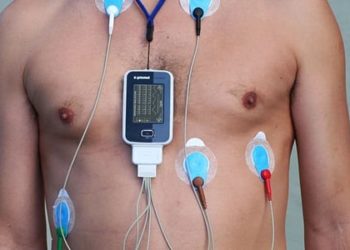
The underdiagnosis of atrial fibrillation is thought to contribute to overall population stroke risk, as patients with atrial fibrillation is known to increase risk for thromboembolic stroke. The population prevalence of atrial fibrillation and the efficacy of population screening is unknown. In this study, investigators report the mass screening of 75 to 76 year old individuals in two Swedish regions by intermittent ECG monitoring over 2 weeks. Over 28 months, 13,331 individuals were invited to be screened, of which 7173 (53.8%) participated. 218 participants (3.0%) were found to have previously undiagnosed atrial fibrillation, of which 37 (17.0%) were diagnosed on first ECG. 666 participants (9.3%) had a previous diagnosis of atrial fibrillation, with an overall atrial fibrillation prevalence of 12.3%. More than 90% of patients with previously undiagnosed atrial fibrillation were started on oral anticoagulation treatment. In total, 3.7% of the screened population was started on oral anticoagulation treatment (including both new diagnoses and previously known diagnoses without treatment). Mass screening efforts for atrial fibrillation in Swedish regions were highly effective in getting participants to start anticoagulation therapy and diagnose atrial fibrillation.
Image: CC
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.




![Neighborhood environments may influence incidence of diabetes [MESA study]](https://www.2minutemedicine.com/wp-content/uploads/2015/06/tai-chi-75x75.jpg)