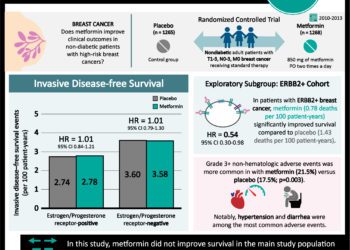
70-gene signature test may accurately determine prognosis in early stage breast cancer
1. In women with early stage breast cancer and discordant genomic and clinical risk, the 5-year survival rate without distant metastasis was 94.7%.
2. Approximately 46% of women with breast cancer who are at high clinical risk might not require chemotherapy.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Women with early stage breast cancer are chosen carefully for treatment with adjuvant chemotherapy. These decisions are based on tumour and patient characteristics. Algorithms have been made to aid in this process but do not take into consideration genetic tumour characteristics. The 70-gene signature (MammaPrint) test classifies tumours into those with good or poor prognosis. This randomized phase-3 study sought to provide evidence for the clinical utility of the addition of the 70-gene signature to standard clinical-pathological criteria in selecting patients for adjuvant chemotherapy.
In women with early stage breast cancer and discordant genomic and clinical risk (low genomic, high clinical risk) the 5-year survival rate without distant metastasis was 94.7% in those who did not receive chemotherapy. There was a higher absolute difference in survival rates between patients who did not receive chemotherapy compared to those who did. There were similar rates of survival in patients who had estrogen-receptor-positive, human epidermal growth factor receptor 2-negative and either node-negative or node positive disease. Strengths of this study include good follow-up for 5 years post-diagnosis and study of a novel genetic test. Further follow-up would be required to determine if the 70-gene signature test impacts mortality.
Click to read the study, published today in NEJM
Relevant Reading: Tailoring therapies—improving the management of early breast cancer: St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2015
In-Depth [randomized controlled trial]: This international, prospective, randomized phase 3 study was conducted from 2007 to 2011 in 9 European countries. Women were between the ages of 18 and 70 with histologically confirmed primary invasive breast cancer with negative lymph nodes until 2009 where they allowed women with up to 3 axillary lymph nodes. The 70-gene signature and the Adjuvant Online was used to determine genomic and clinical risk respectively. Low clinical risk was defined as 10-year probability of breast-cancer-specific survival without systemic therapy of >88% or >92% for ER positive or ER negative tumours. Discordant risk was defined as high clinical risk and low genomic risk, or low clinical risk and high genomic risk. The primary end point was survival without distance metastasis (event-free rate at 5 years) as assessed in a time-to-event analysis. Secondary end points included proportion of patients who received chemotherapy according to clinical and genomic risk.
A total of 1550 patients (23.3%) were deemed high clinical risk but low genomic risk. At 5 years, the rate of survival without distant metastasis in the high clinical risk but low genomic risk (94.7%, 95%CI 92.5-96.2). The absolute difference in survival rate for these patients on chemotherapy vs. no chemotherapy was 1.5%. Similar rates of survival were reported in all cancer subgroup analyses.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.