Active surveillance in favorable-risk prostate cancer associated with low long-term mortality
1. In a large prospective cohort study of approximately 1300 men with favorable-risk prostate cancer that underwent active surveillance, there was a low incidence of prostate cancer progression and mortality at 15 years follow-up.
Evidence Rating Level: 2 (Good)
Study Rundown: The lifetime risk of developing prostate cancer in the United States is 1 in 7 men. Active surveillance (AS) has previously demonstrated similar disease-specific survival rates compared to immediate curative intervention in patients with low-risk prostate cancer. However, there is limited data on the long-term outcome of patients on AS. The purpose of this study was to provide long-term survival data in a large prospective cohort of men with favorable-risk prostate cancer receiving AS.
The study prospectively recruited approximately 1300 men with low- or very low-risk prostate cancer on AS monitoring, which comprises of semi-annual serum prostate specific antigen (PSA), digital rectal exam, as well as annual prostate biopsy. At the conclusion of the trial, the overall and cancer-specific survival rate of the AS cohort were 93% and 99.9% at 10-years and 69% and 99.9% at 15-years follow-up. Similar rates were observed for metastasis-free survival. Furthermore, curative intervention due to disease progression was only required for 50% of patients at 10-years follow-up. The results of this study support the hypothesis that AS can be used as a safe management approach in patients with favorable-risk prostate cancer. The study is strengthened by the large sample size; however, it should be noted that that the population enrolled was primarily Caucasian, which may limit the generalizability of this study to other racial groups. Nevertheless, this study adds to the growing body of evidence supporting the option of AS for men with low-risk prostate cancer.
Click to read the study in JCO
Click to read an accompanying editorial in JCO
Relevant Reading: Long-term follow-up of a large active surveillance cohort of patients with prostate cancer
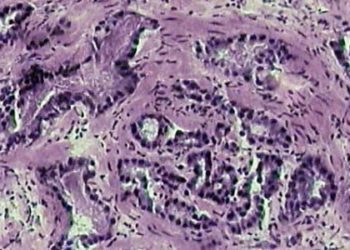
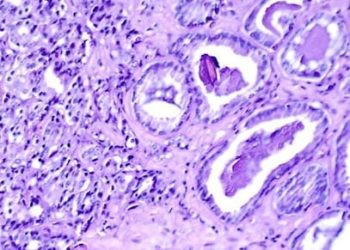
In-Depth [prospective cohort study]: Beginning in 1995, a total of 1298 men with favorable risk prostate cancer receiving AS were enrolled in the study. Favorable-risk prostate cancer was defined as clinical very low-risk (VLR) and low risk (LR) prostate cancer. VLR includes clinical stage T1c disease, prostate-specific antigen (PSA) density (PSAD) less than 0.15ng/mL, biopsy Gleason score ≤6, two or fewer positive biopsy cores, and a maximum of 50% involvement of any biopsy core with cancer. LR was defined as clinical stage ≤T2a, PSA <10ng/mL, and Gleason score ≤6. The active surveillance protocol involved semiannual PSA measurement and digital rectal examination, and annual 12- to 14-core biopsy. When repeat biopsies no longer meeting inclusion criteria, the patients’ disease was reclassified and curative intervention was recommended. Primary outcomes were overall, cancer-specific, and metastasis-free survivals, and secondary outcomes rates of biopsy reclassification. Kaplan-Meier time-to-event curves and multivariable regression analysis were used. The median age of the cohort was 66 years old; 88% of the patients enrolled were Caucasian and 7.4% were African-American. Overall, cancer-specific, and metastasis-free survival rates were 93%, 99.9%, and 99.4% at 10 years follow-up, and 69%, 99.9% and 99.4% at 15 years follow-up. Biopsy reclassification cumulative incidence was 26% and 31% and 10- and 15-years follow-up, respectively.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.