Anti-interleukin-17A therapy shows promise in psoriatic arthritis
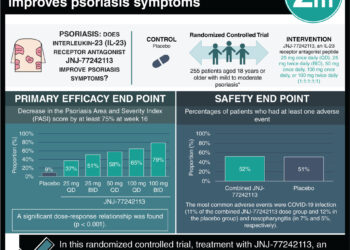
1. Among patients with psoriatic arthritis, secukinumab was superior to placebo at week 24 in terms of ACR20 response, which captures a 20% decrease in number of tender and swollen joints and clinically relevant physician and patient reported outcomes.
2. Adverse events including myocardial infarction and stroke were more common in the secukinumab groups, though the study was not large or long enough to adequately assess these outcomes.
Evidence Rating Level: 1 (Excellent)
Study Rundown: The treatment of patients with psoriatic arthritis was greatly augmented by tumor necrosis factor (TNR) inhibitors. However, there are some patients whose symptoms do not resolve with these agents. Previous studies had shown that inhibition of the interleukin-17A receptor, thought to be essential in the pathogenesis of psoriatic arthritis, was associated with improved signs and symptoms of psoriatic arthritis. In this double-blind, phase 3 study 606 patients with psoriatic arthritis were randomized to receive either intravenous secukinumab, an anti-IL-17A monoclonal antibody in both intravenous and subcutaneous forms (at either 150mg or 75mg every 4 weeks), or a placebo.
There were significantly better response rates in terms of the American College of Rheumatology (ACR) 20, a composite measure capturing number of tender/swollen joints as well as patient’s and physician’s global assessment of disease over 24 weeks of placebo control. Infections (especially candida infections), MIs, and strokes were more common in the secukinumab treatment groups. This study draws strength from its randomized design. Further studies that are both larger and have a longer follow-up period are needed to investigate its long-term efficacy and safety profile.
Click to read the study, published today in NEJM
Relevant Reading: Secukinumab in plaque psoriasis – results of two phase 3 trials
In-Depth [randomized controlled trial]: This study involved 606 patients randomized in a 1:1:1 ratio to receive IV secukinumab (10mg/kg) at 0, 2, and 4 weeks followed by subcutaneous secukinumab at a dose of either 150mg or 75mg every 4 weeks, or placebo. The primary end point was the ACR20, which is a composite measure of 20% improvement from baseline in the number of tender and swollen joints and other patient and physician-reported measures. Secukinumab was superior to placebo at week 24 in terms of ACR20 response (50.0% and 55.0% in the 150mg and 75mg groups, respectively, compared to 17.3% in the placebo group, P<0.001); the same held for more rigorous ACR50 and ACR70 results. These findings also held true through 52 weeks of therapy in the extended follow-up period. The rate of myocardial infarction was 0.3 per 100 patient-years in the secukinumab vs 0 cases in the placebo group; the combined rate for stroke was 0.6 per 100 patient-years in the secukinumab groups versus 0 cases in the placebo group.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.