Atezolizumab and nab-paclitaxel prolong progression-free survival for patients with metastatic triple-negative breast cancer: the IMpassion130 trial
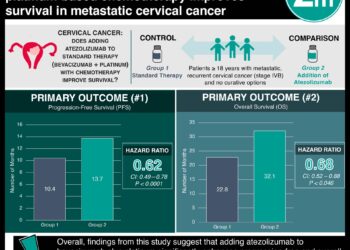
1. This randomized controlled trial of women with metastatic triple-negative breast cancer reports that patients who were received atezolizumab with nab-paclitaxel had longer periods of progression-free survival compared to patients who received placebo plus nab-paclitaxel therapy.
2. Rates of adverse events were comparable in both study groups, however it was noted that the incidence of grade 3 or 4 adverse events and immune-related averse events were higher in the atezolizumab plus nab-paclitaxel group.
Evidence Rating Level: 1 (Excellent)
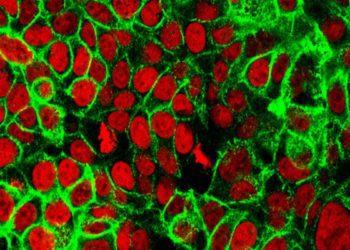
Study Rundown: Triple-negative breast cancer is generally regarded as more aggressive and difficult to treat as compared to other subtypes of breast cancer, ultimately leading to poor survival outcomes among patients. The use of immunotherapy with chemotherapy has been investigated in the context of various types of cancers as a way to augment chemotherapy treatment responses. In this current analysis, the IMpassion130 phase three randomized control trial investigated the efficacy of the combined use of atezolizumab, an immunologic agent that binds to programmed death ligand 1 (PDL1) to prevent T-cell suppression with nab-paclitaxel to assess their combined impact on cancer progression-free survival and overall survival as compared to a placebo plus nab-paclitaxel regimen. The safety profiles of each arm were also assessed. Overall, authors found that the combination of atezolizumab with nab-paclitaxel therapy led to longer cancer progression-free survival periods compared to placebo plus nab-paclitaxel. Results were consistent both among their intention-to-treat population and among patients who had PDL1 positive tumors. While preliminary, these results present evidence that immunotherapy could be considered as a possible adjunct therapy for patients with metastatic triple negative breast cancer.
Strengths of this study include evaluation of patient PD-L1 positive patients in addition to additional extensive subgroup analysis. The study lacked author disclosed or observed limitations and lack of non-survival based outcomes, such as quality of life or other patient functional outcomes.
Click to read the study in NEJM
Relevant Reading: Beyond melanoma: inhibiting the PD-1/PD-L1 pathway in solid tumors
In-Depth [randomized controlled trial]: This phase three, multicenter randomized controlled trial enrolled 902 patients across 41 countries between Jun 2015 to May 2017. Patients were randomized in a 1:1 ratio to either receive atezolizumab plus nab-paclitaxel (n=451) or placebo plus nab-paclitaxel therapy (n=451). Treatment regimens were based off of 28-day cycles; in the absence of disease progression, patients were scheduled to receive at least 6 cycles of chemotherapy. Atezolizumab or placebo were administered at doses of 840mg on days 1 and 15 of each cycle. Nab-paclitaxel was administered at a dose of 100mg/m2 on days 1, 8 and 15. The primary outcomes of this analysis were to evaluate for progression-free survival and overall survival rates. Survival outcomes were also assessed within a subgroup of patients who were PD-L1 positive (n=369 patients; n=185 atezolizumab with nab-paclitaxel; n=184 placebo with nab-paclitaxel). The median follow-up period was 12.9 months among the total cohort (13.0 months in the atezolizumab-nab-paclitaxel group; 12.5 months in the placebo-nab-paclitaxel group). Progression free survival was noted to be significantly longer in the atezolizumab and nab-paclitaxel group compared to placebo (median survival 7.2 months vs 5.5 months; hazard ratio [HR], 0.80; 95% confidence interval [CI], 0.69 to 0.92; P=0.002). Prolonged progression-free survival was also observed among patients who were in the PD-L1 positive subgroup treated with atezolizumab plus nab-paclitaxel (median progression-free survival 7.5 months vs 5.0 months, HR: 0.62; 95% CI: 0.49 to 0.78, P<0.001). From a safety standpoint, adverse events occurred in 99.3% of patients in the atezolizumab plus nab-paclitaxel group compared to 97.9% in the placebo group. The most common adverse effects included neutropenia, peripheral neuropathy, fatigue and anemia. The rate of grade 3 or 4 adverse events in the atezolizumab therapy group were 48.7% and 42.2% in the placebo group.
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.