Bioresorbable vascular scaffolds linked to increased risk of adverse events at mid- and long-term
1. In comparison to everolimus-eluting metallic stents (EESs), bioresorbable vascular scaffolds (BVSs) increased the chances for thrombotic events at both mid- and long-term follow-up.
2. Compared to patients with EESs, those with BVSs had a heightened risk (that increased over time) of myocardial infarction, target lesion revascularization (TLR), and target lesion failure (TLF).
Evidence Rating Level: 1 (Excellent)
Study Rundown: Compared to bare-metal stents, drug-eluting stents decrease the frequency of in-stent stenosis, TLR, and myocardial infarction for people undergoing percutaneous coronary interventions (PCIs). However, there are still concerns that these drug-eluting stents may be associated with late and very late stent thrombosis. BVSs were designed to combat these problems, but recent data suggests that BVSs may be associated with a higher risk of cardiovascular events compared to EESs. This study sought to estimate how often scaffold thrombosis occurred after BVS implantation and to compare everolimus-eluting BVSs with EESs regarding safety and efficacy in adults who had a PCI. Through a systematic review and meta-analysis, researchers found that in comparison to EESs, BVSs increased the chances for thrombotic events, including scaffold or stent thrombosis, at both mid- and long-term follow-up. The authors suggest that to improve clinical outcomes of BVSs, better scaffold-specific techniques and scaffold designs are necessary.
Considering that long-term safety and efficacy data were not clearly established in previous trials, a strength of this study is that it evaluates mid- and long-term follow-up data. Limitations of the study include unclear quality of the observational studies used and that some data were not published.
Click to read the study in Annals of Internal Medicine
Click to read an accompanying editorial in Annals of Internal Medicine
Relevant Reading: Everolimus-eluting bioresorbable vascular scaffolds versus everolimus-eluting metallic stents: a meta-analysis of randomised controlled trials
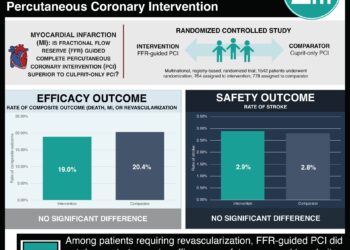
In-Depth [systematic review and meta-analysis]: Using PubMed, the Cochrane Library, EMBASE, conference proceedings, and pertinent Web sites, researchers completed a meta-analysis of 7 randomized controlled trials (RCTs) and 38 observational studies that included adults with coronary artery disease who received a BVS or an EES and reported thrombosis or other outcomes such as myocardial infarction, revascularization, or death. At a median follow-up of 1 year and beyond 1 year, the rate of scaffold thrombosis following BVS implantation was 1.8% and 0.8%, respectively. In 7 trials with a direct comparison of BVSs with EESs, there was an increased risk (odds ratio [OR], 3.40 [CI, 2.01 to 5.76]) of scaffold thrombosis with BVSs at 25 months (median follow-up). Compared to patients with EES, those with BVSs had a heightened risk of myocardial infarction (OR, 1.63), TLR (OR, 1.31), and TLF (OR, 1.37). Over time, the odds for these three risk factors all increased.
Image: PD
©2017 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.