Broadly neutralizing antibody treatment does not prevent overall HIV-1 acquisition
1. Broadly neutralizing antibody treatment was shown not to prevent overall HIV-1 acquisition compared to placebo.
2. Sub-group analysis demonstrated broadly neutralizing antibodies could prevent acquisition in HIV-1 isolates.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Though risk reduction measures have helped blunt the spread of human immunodeficiency virus (HIV), preventive treatments can help increase the effectiveness of prevention. VRC01 is an immunoglobulin G-1 (IgG1) broadly neutralizing antibody (bnAb) directed at HIV type 1 (HIV-1). This study determined whether VRC01 was capable of preventing HIV-1 infection. VRC01 provided to at-risk cis-gendered men and trans-gendered persons in the Americas and Europe, and at-risk women in sub-Saharan Africa did not prevent overall HIV-1 acquisition compared to placebo. In prespecified analyses pooling data across trials, the incidence of infection with VRC01 sensitive isolates was lower amongst VRC01 recipients compared to placebo. Study limitations include the lack of reporting on long-term outcomes after treatment with bnAb. Nonetheless, this study’s results are significant as a proof-of-concept trial. Despite showing bnAb did not prevent HIV-1 acquisition, it did show that bnAb could be used against susceptible HIV-1 isolates.
Click to read the study in NEJM
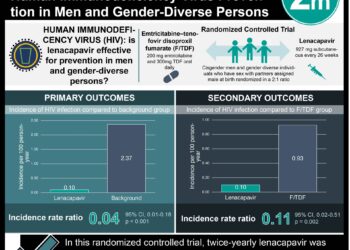
In-Depth [randomized controlled trial]: This study reported the results on two parallel phase 2b, randomized, proof-of-concept efficacy trials of bnAb VRC01 in at-risk cis-gendered men and trans-gendered persons in the Americas and Europe, and at-risk women in sub-Saharan Africa. 2,699 participants (HVTN 704/HPTN 085 trial) and 1,924 participants (HVTN 703/HPTN 081 trial) were enrolled in the study and randomized in a 1:1:1 ratio to either low dose VRC01 (10 mg/kg), high dose VRC01 (30 mg/kg) or placebo treatment, respectively. The study’s primary endpoint was documental HIV-1 infection by the week 8 trial visit. In HVTN 704/HPTN 085, the overall estimated prevention efficacy was 22.4% compared to placebo at the week 80 visit in the low dose group (95% confidence interval [CI], −25.5 to 52.0), 30.9% (95% CI, −13.9 to 58.0) in the high dose group, and 26.6% (95% CI, −11.7 to 51.8; P = 0.15) in the pooled VRC01 group. In HVTN 703/HPTN 081, the overall estimated prevention efficacy was –9.3% (95% CI, −85.3 to 35.5) in the low-dose group, 27.0% (95% CI, −30.7 to 59.3) in the high-dose group, and 8.8% (95% CI, −45.1 to 42.6; P = 0.70) in the pooled VRC01 group. In assessing the association between protection at week 80 and viral strain sensitivity, an analysis of pooled data from the two trials showed the incidence of infection with sensitive viruses was 0.20 per 100-person years amongst VRC01 recipients compared to 0.86 per 100-person years in placebo recipients (estimated prevention efficacy, 75.4%; 95% CI, 45.5 to 88.9). Overall, this study demonstrated bnAb did not prevent overall HIV-1 acquisition but did show proof of concept efficacy in bnAb sensitive HIV-1 isolates.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.