Brodalumab may be effective for difficult-to-treat psoriasis
1. Treatment with brodalumab was shown to be safe and efficacious in patients with psoriatic arthritis or who previously failed treatment with biologic therapy.
Evidence Rating Level: 2 (Good)
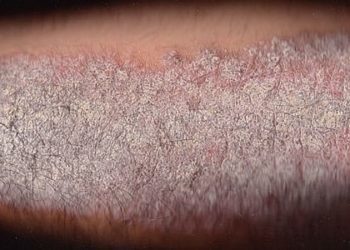
Study Rundown: Psoriasis is a chronic inflammatory skin disease that classically presents with itchy red lesions with a silvery scale. A difficult-to-treat subgroup of these patients, those with a history of psoriatic arthritis (PsA) or those who have failed biologic therapy, necessitate more treatment options. These authors performed a secondary analysis on the safety and efficacy of brodalumab, a novel therapy previously shown to improve psoriatic outcomes, in a difficult-to-treat cohort. When comparing patients with or without history of biologic use and patients with or without self-reported PsA, there were similar improvements in outcome and quality of life as compared to those treated with a placebo, indicating the utility of the therapy even for patients classically considered to have difficult-to-treat disease. While the randomized-controlled double-blind, placebo-controlled design is inherently strong, the study had notable limitations: (1) the differences between the subgroups were not compared statistically, (2) history of psoriatic arthritis was self-reported, and (3) information regarding reason for discontinuation of biologic therapy was not collected.
Click to read the study in JAAD
Relevant Reading: Strategies to maximize treatment success in moderate to severe psoriasis: establishing treatment goals and tailoring of biologic therapies
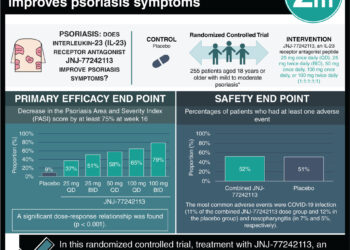
In-Depth [randomized controlled trial]: Four separate measures assessing quality of life and psoriasis severity were used to evaluate the two subsets of patients with moderate-severe difficult-to-treat plaque psoriasis [(1) those with or without PsA and (2) with or without history of biologic use]. Among the sub-groups, the mean duration of psoriasis ranged from 17.3 years to 24.3 years. Within all sub-groups, there were similar trends of 75%, 90%, and 100% improvement at 12 weeks when compared to baseline using the Psoriasis Area and Severity Index score (PASI) in those that received either 140 mg or 210 mg of medication as compared to those that received placebo with one exception. In the 140 mg group, the percentage of patients with 100% improvement was observed to be higher in patients with a history of PsA. Analyzing the sub-groups using the Dermatology Life Quality Index (DLQI), Psoriasis Symptom Inventory (PSI) and Physician Global Assessment Scales (PGA) demonstrated similar results. The amount of adverse events in patients who received brodalumab as compare to placebo was comparable amongst all subgroups.
More from this author: Tech-aided surveillance of patients at high risk for melanoma aids early diagnosis, Indoor tanning linked to high skin cancer risk in US and abroad, Free drug samples may alter prescription habits of dermatologists, Atopic dermatitis may be more persistent than previously understood
Image: PD
©2014 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. No article should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2 Minute Medicine, Inc.