Improvements in therapeutic and managing standards associated with significant reduction in risk of persistent disability in patients with pediatric-onset multiple sclerosis
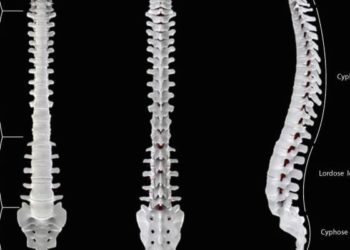
1. In recent years, the risk of persistent disability has been significantly reduced in patients with pediatric-onset multiple sclerosis (POMS), likely owing to availability of new disease-modifying therapies (DMTs) and changes to therapeutic paradigms over time.
2. Further research in therapeutic efficacy and management in the pediatric population is needed in order to improve the prognosis of patients with POMS.
Evidence Rating Level: 2 (Good)
Study Rundown: Treatment of pediatric-onset multiple sclerosis (POMS) is largely based on the use of disease-modifying therapies (DMTs) originally tested in the adult population. In recent years, the availability of new DMTs and changes in therapeutic paradigm have led to a general improvement of multiple sclerosis (MS) prognosis in adults. However, it remains unclear whether this improvement applies to pediatric patients, whose early management is often more challenging. This multicenter retrospective study using data from the Italian MS Registry in May 2019 sought to evaluate whether the disease course of POMS has changed over time in association with improvements in therapeutic and management standards. Specifically, the study compared time to reach disability milestones from MS clinical onset in a large patient cohort divided into four consecutive diagnosis epochs (<1993, 1993-1999, 2000-2006, and 2007-2013). The primary outcome was the time to reach certain pre-set disability milestones, assessed via the Expanded Disability Status Scale (EDSS) score at clinical evaluation and upon the last available visit. Among 3,198 patients enrolled with POMS, the median survival time to reach a EDSS score of 4.0 and 6.0 was 31.7 and 40.5 years, respectively. Over the period of four diagnosis epochs, the cumulative risk of reaching disability milestones gradually decreased – both for EDSS score of 4.0 and 6.0. This was likely associated with a greater number of patients treated with DMTs, especially using high-potency drugs, that were also given earlier and for a longer period in patients from later epochs. Thus, the risk of persistent disability in patients with POMS has been reduced by 50% to 70% in recent diagnosis epochs, likely owing to the availability of new DMT and shifts in therapeutic paradigms. A limitation of this study, however, was that due to the particular retrospective design, one cannot definitively exclude other possible confounding causes for the reduction in disability risk, including general improvements in health care, early diagnosis, clinical assessment, and other therapeutic interventions (e.g., physiotherapy).
Click to read the study in JAMA Neurology
Relevant Reading: Real-world effectiveness of initial disease-modifying therapies in pediatric multiple sclerosis
In-Depth [retrospective cohort]: This retrospective, multicenter, observational study used data extracted from the Italian MS Registry in May 2019, where information on over 59,000 MS patients were collected. Upon screening via inclusion/exclusion criteria, 3,198 patients were selected from 4,704 eligible patients with POMS (69% female). The mean age at onset was 15.2 years and the median time to diagnosis was 3.2 years. The annual relapse rate at one and three years of disease progression was 1.3 and 0.6 respectively, with a mean (SD) follow-up of 21.8 (11.7) years. The median survival time to reach a EDSS score of 4.0 and 6.0 was 31.7 and 40.5 years respectively. When comparing the cumulative risk of reaching disability milestones over time, analysis showed a gradual decrease over four diagnosis epochs. This was consistent for both an EDSS score of 4.0 (HR, 0.70; 95%CI, 0.58-0.83 in 1993-1999; HR, 0.48; 95%CI, 0.38-0.60 in 2000-2006; HR, 0.44; 95%CI, 0.32-0.59 in 2007-2013) and 6.0 (HR, 0.72; 95%CI, 0.57-0.90; HR, 0.44; 95%CI, 0.33-0.60; HR, 0.30; 0.20-0.46). The study concluded that this trend among later diagnosis epochs is likely owing to a greater number of patients with POMS treated with DMTs, especially using high-potency drugs, that were also administered earlier and for a longer period. Statistical analysis showed that demographic characteristics and clinical disease activity at onset did not change significantly over time.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.