Ivosidenib and azacitidine provide significant benefits in IDH1-mutated acute myeloid leukemia
1. Compared to placebo and azacytidine, the combination of ivosidenib and azacitidine demonstrated significant clinical benefit for IDH1-mutated acute myeloid leukemia.
2. Ivosidenib and azacitidine were associated with a higher probability of having event-free survival compared to the placebo and azacytidine group for IDH1-mutated acute myeloid leukemia.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Acute myeloid leukemia affects primarily older adults, with a median age of 68 years. isocitrate dehydrogenase 1 (IDH1)-mutated acute myeloid leukemia is associated with older age and a poorer prognosis. Ivosidenib, a small-molecule inhibitor of mutant IDH1, has been shown to have clinical activity in patients with newly diagnosed IDH1-mutated acute myeloid leukemia. However, there is a gap in knowledge as to understanding the clinical benefits of ivosidenib and azacitidine combination therapy, as ivosidenib monotherapy is already approved in the US for adults with relapsed, refractory, or newly diagnosed IDH1-mutated acute myeloid leukemia who are 75 years of age or older or who have coexisting conditions that preclude chemotherapy. This study found that ivosidenib and azacitidine significantly provided clinical benefit for newly diagnosed IDH1-mutated acute myeloid leukemia, leading to improved event-free survival, response, and overall survival. This study was limited by fewer patients recruited to the trial than initially planned, resulting in limited data interpretation in certain preplanned subgroup analyses. Nevertheless, these study’s findings are significant, as they demonstrate that ivosidenib and azacitidine offer significant clinical benefits in patients with newly diagnosed IDH1-mutated acute myeloid leukemia who were ineligible for induction chemotherapy.
Click to read the study in NEJM
Relevant Reading: Durable Remissions with Ivosidenib in IDH1-Mutated Relapsed or Refractory AML
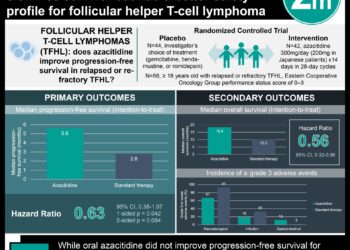
In-Depth [randomized control trial]: This global, double-blind, randomized, placebo-controlled, phase 3 trial studied the efficacy and safety of ivosidenib and azacitidine as compared to placebo and azacytidine, in 146 patients. Patients who were 18 years or older, with a centrally confirmed diagnosis of previously untreated IDH1-mutated acute myeloid leukemia were eligible for the study. Patients who had been previously treated with an IDH1 inhibitor or hypomethylating agent or with inadequate hepatic and renal function were excluded from the study. The primary outcome measured was event-free survival, defined as the time from randomization until treatment failure, relapse from remission, or death from any cause, whichever occurred first. Outcomes in the primary analysis were assessed via the intention-to-treat population with Cox proportional hazards model, a log-rank test, and a Cochran-Mantel-Haenszel test with stratification factors according to geographic region and disease status. Based on the analysis, at a median follow-up of 12.4 months, event-free survival was significantly longer in the ivosidenib and azacitidine group compared to the placebo and azacitidine group (hazard ratio for treatment failure, relapse from remission, or death, 0.33; 95% Confidence Interval [CI], 0.16 to 0.69). The probability that a patient would remain event-free at 12 months was 37% in the ivosidenib and azacitidine group and 12% in the placebo and azacitidine group. The median overall survival was 24 months with ivosidenib and azacitidine and 7.9 months with placebo and azacitidine (hazard ratio for death, 0.44; 95% CI, 0.27 to 0.73). Overall, this study demonstrated that ivosidenib and azacitidine provided significant clinical benefit in terms of event-free survival, response, and overall survival compared to placebo and azacitidine in patients with newly diagnosed IDH1-mutated acute myeloid leukemia who were ineligible for induction chemotherapy.
Image: PD
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.

![Addition of bevacizumab fails to benefit overall survival in newly diagnosed glioblastoma [RTOG 0825 trial]](https://www.2minutemedicine.com/wp-content/uploads/2014/02/AFIP-00405558-Glioblastoma-Radiology-e1381685577283-350x250.jpg)