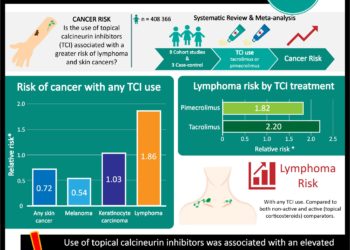
New, slow-release Tacrolimus may be better for kidney transplants
1. Once-daily doses of slow-release formulation Tacrolimus (1xSRT) showed similar treatment efficacy when compared to standard twice-daily doses of Tacrolimus (2xT).
2. 1xSRT treatment required lower doses of Tacrolimus to achieve the similar trough levels (lowest level of the drug present in the patient) when compared to 2xT.
Evidence Rating Level: 2 (Good)
Study Rundown: Transplant rejection is a major complication associated with kidney transplantation, and post-surgery treatment with the drug, Tacrolimus, has been shown to prevent acute rejection. Currently, Tacrolimus is administered as a twice-daily pill. Unfortunately, multiple-per-day dosing is linked to increased risk of patient nonadherence, which in turn may lead to acute rejection and graft failure.
This study examined the efficacy of a slow-release formulation of Tacrolimus that allows the treatment to be administered once daily. Proprietary technology was used to improve the drug delivery system. When examining the incidence of death, graft failure, and treatment safety, the 1xSRT was as effective as the 2xT. In several measures, the 1xSRT treatment actually appeared more effective, though this was not statistically significant. This study was unable to evaluate the additional benefit 1xSRT treatment may provide as a result of increased patient compliance, because 1xSRT patients were give a second daily placebo dose to maintain patient blindness to treatment type. This study provides evidence that a single daily dose of Tacrolimus (in slow-release form) may be as therapeutically effective as a twice-daily dose, suggesting an opportunity to increase patient adherence in critical post-transplant treatments. The authors of this study disclose multiple conflicts of interest.
Click to read the study in the American Journal of Transplantation
Relevant Reading: Conversion from twice daily tacrolimus capsules to once daily extended-release tacrolimus (LCP-Tacro): Phase 2 trial of stable liver transplant recipients
In-Depth [prospective, randomized, Phase III study]: This study examines the efficacy of 1xSRT treatment of 206 kidney transplant patients when compared with the conventional 2xT treatment of 219 kidney transplant patients. Following initial dosing, levels of Tacrolimus in the blood were measured, and treatment doses were adjusted to maintain patient trough levels within target ranges of 6-11 ng/ml for the first 30 days of study, and 4-11 ng/ml for the rest of the trial. The primary endpoints were events of treatment failure including death, graft failure, and biopsy-proven acute rejection. Safety evaluations collected the incidences of adverse events, serious adverse events, and discontinuation due to adverse events. Follow-up from the time of treatment randomization was 12-months.
Treatment failure was 18.3% for 1xSRT patients, and 19.6% for 2xT patients, with no significant difference between mortality, graft failure, or acute rejection events. Adverse events were also not significantly different between the two study groups, though 1xSRT treatment had a slightly lower number of severe adverse events. 1xSRT patients required a daily dose that was 14% lower than the dose required to maintain similar trough levels in the 2xT patients.
More from this author: Deep brain stimulation linked to declining brain function in Parkinson’s patients, Donor and recipient regulatory T-cells may promote transplant tolerance in mice, Growth factor embedded film promotes robust bone repair, The 3C Study: Alemtuzumab may reduce acute rejection following kidney transplant, Paclitaxel-coated balloons may decrease bronchial stenosis in lung transplants
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.