Nicotine biomarker predicts rates of smoking cessation in response to varenicline or nicotine patch
1. Varenicline treatment was shown to provide greater quit rates than nicotine patches in normal nicotine metabolizers but was not more effective in slow nicotine metabolizers.
2. Varenicline treatment had a greater side effect profile in slow nicotine metabolizers than normal nicotine metabolizers.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Smoking has remained a large health problem despite efforts over the past several decades to promote abstinence and cessation. Presently, behavior modification and pharmacologic treatment are the forerunners in assisting patients to quit smoking. The nicotine metabolite ratio (NMR, 3’-hydroxycotinine:cotinine) has recently arisen as a biomarker for identifying patients that may potentially respond to various pharmacologic therapies, with regards to their ability to metabolize nicotine. Using this knowledge, the authors undertook a randomized, placebo-controlled trial to examine the use of varenicline vs. nicotine patch therapy in patients identified as slow or normal nicotine metabolizers by the NMR.
The results of the study showed that varenicline was more effective than the nicotine patch at aiding in smoking cessation in normal nicotine metabolizers, but had equal efficacy in slow metabolizers. Additionally, varenicline had a greater side effect profile in slow metabolizers than normal metabolizers compared to placebo. Strengths of this study included the fact that it was the largest of its kind to examine pharmacologic therapy as related to NMR utilization. Additionally, although the efficacy of bupropion has been examined with regards to NMR this was the first study where varenicline had been investigated. This study was limited by strict exclusion criteria imposed on the patient selection process including absence of major medical or psychiatric illnesses, which may have greatly limited the generalizability of the results to healthy patients free of comorbidities.
Click to read the study in The Lancet
Relevant Reading: Nicotine metabolite ratio predicts efficacy of transdermal nicotine for smoking cessation
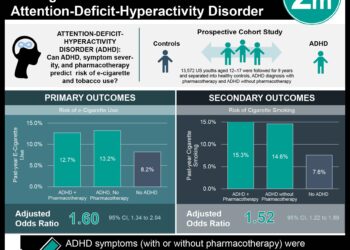
In-Depth [randomized controlled trial]: This study included 1,246 patients randomized based on their initial NMR status as either normal (n=584) or slow (n=662) metabolizers into three groups for 11 weeks of treatment: 1) placebo (placebo pill and patch, n=408), 2) nicotine patch (placebo pill, active patch, n=418), and 3) varenicline (varenicline pill, placebo patch, n=420). The primary outcome of this study was 7 day abstinence at the end of 11 week treatment while secondary endpoints included 6 and 12 month abstinence, as well as side-effect and withdrawal symptoms.
The results showed that varenicline was more effective than nicotine patch at aiding in smoking cessation in normal metabolizers (Odds Ratio [OR] 2.17, 95% CI 1.38-3.42; p=0.001), but not in slow metabolizers (OR 1.13, 0.74-1.71: p=0.56) at the end of treatment, with similar results at 6 months. Additionally, varenicline as opposed to placebo had a significantly greater side effect profile for slow metabolizers (β=0.61, 95% CI -0.10 to 1.32; p=0.09) than normal metabolizers (β=-0.44, 95% CI -1.19 to 0.30; p=0.24). Serious adverse effects were observed in 16 (3.9%) of the placebo group, 22 (5.3%) of the nicotine patch group, and 11 (2.6%) of the varenicline group.
More from this author: Ebola and Marburg vaccines safe and well tolerated in Phase 1b trial in Uganda; Sanitation efforts may not be effective at preventing fecal exposure in India; Increased Vitamin D concentrations may not decrease type 2 diabetes risk; Benralizumab does not reduce the rate of acute COPD exacerbations; Lifetime diabetes risk rises as incidence increases and mortality decreases
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. No article should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2 Minute Medicine, Inc.