Nivolumab improves survival in head and neck squamous cell carcinoma: The CheckMate 141 trial
1. Nivolumab significantly increased median overall survival in patients with platinum refractory relapsed metastatic head and neck squamous cell carcinoma (SCC) compared to standard therapy.
2. Compared with standard chemotherapy options, nivolumab was associated with fewer grade 3 or 4 adverse effects and a higher patient-reported quality of life.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Head and neck cancer is the seventh most common type of cancer worldwide and has a notably poor prognosis in the metastatic stage. Nivolumab is in a new class of agents termed programmed death 1 inhibitors, which use the body’s immune system to recognize and combat cancer. This study assessed the efficacy and safety of nivolumab against standard chemotherapy in platinum refractory relapsed head and neck squamous cell carcinoma (SCC). The primary end point – overall median survival – was significantly increased in the nivolumab group for all patient populations. Among secondary endpoints, nivolumab demonstrated significantly fewer serious adverse events, higher response rates, and improved patient-reported quality of life compared to standard therapy.
This was a randomized, phase III clinical trial that demonstrated an immunotherapeutic response for head and neck cancer. Head and neck cancers are characteristically difficult to treat and have poor survival rates, making nivolumab a potentially breakthrough treatment option. However, nivolumab displays a different magnitude of effect in individual cases and additional research on biomarkers and biological criteria to identify patients most likely to benefit should be conducted.
Click to read the study, published in NEJM
Relevant Reading: Nivolumab plus ipilimumab in advanced melanoma
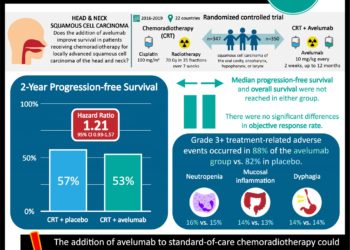
In-Depth [randomized controlled trial]: This randomized trial assigned 361 patients to nivolumab (n = 240) or standard therapy (n = 121; physician’s choice of methotrexate in 46 patients, docetaxel in 52 patients, or cetuximab in 13 patients). The primary endpoint was overall survival from time of randomization. Secondary endpoints were progression-free survival, objective response rate according to RECIST version 1.1, associations of survival and PD-1 level and human papillomavirus (HPV) status (via p16 expression), survival, and patient-reported quality of life assessed through clinically validated questionnaires.
Median overall survival was 7.5 months in the nivolumab group vs. 5.1 months in patients receiving standard therapy (HR 0.70; 97.73%CI 0.51 to 0.96; p = 0.01). No significant difference was found between the rate of progression-free survival between treatments (HR 0.89; 95%CI 0.70 to 1.13; p = 0.32). The response rate in the nivolumab group was 13.3% with 6 complete and 26 partial responses, compared to 5.8% with 1 complete and 6 partial responses in the standard therapy group. In patients with HPV positive disease, median overall survival was 9.1 months with nivolumab compared to 4.4 months with standard therapy. In patients with PD-1 expression greater than 1% the HR was 0.55 compared to 0.89 in those with an expression level less than 1%. Patients treated with standard chemotherapy reported a worsening of physical, role, and social functioning that remained stable in the nivolumab group. Treatment related adverse events of any grade were similar between the two groups, but fewer grade 3 or 4 adverse events were reported in the nivolumab group vs. standard therapy (13.1% vs. 35.1% of patients). Adverse events more common to nivolumab included those skin and endocrine related.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc