Predicted long-term effects of dapagliflozin in heart failure suggest association with improved event-free survival
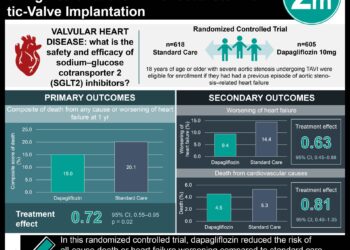
1. A prediction of the long-term effects of dapagliflozin from the short-term data in the Dapagliflozin and Prevention of Adverse-Outcomes in Heart Failure (DAPA-HF) trial suggest it reduces the risk of HF events by 26% and improves event-free survival by 3.0 to 1.2 years depending on the patient’s age compared with placebo.
2. The estimate of long-term effects of dapagliflozin suggests it improves all-cause mortality by 1.7 years compared with placebo.
Evidence Rating Level: 3 (Average)
Study Rundown: The sodium glucose cotransporter 2 inhibitor, dapagliflozin, has been shown in the Dapagliflozin and Prevention of Adverse-Outcomes in Heart Failure (DAPA-HF) randomized clinical trial to improve short-term survival in patients with heart failure and reduced ejection fraction (HFrEF). However, the follow-up on this study was only 16-18 months, so it might underestimate the benefits of life-long dapagliflozin use. This study estimates the lifetime event-free survival time (the time until the first worsening HF event, including an unplanned hospitalization, urgent outpatient consultation for HF, or cardiovascular death) of patients receiving dapagliflozin treatment. The DAPA-HF trial randomized participants to dapagliflozin or placebo in addition to their standard therapy. The data from 4744 HFrEF patients that completed the DAPA-HF trial were used to predict estimates of mean event-free survival time and all-cause mortality beyond the 17.6±5.2 months treatment/follow-up during the original study. Dapagliflozin reduced the risk of HF events by 26% and improved event-free survival at all ages compared with placebo by 3.0 to 1.2 years depending on the patient’s age. Dapagliflozin improved event-free survival of participants who were on monotherapy (only dapagliflozin) and those who were on quadruple therapy (dapagliflozin with angiotensin receptor-neprilysin inhibitor, β-blocker, and mineralocorticoid receptor antagonist). Dapagliflozin decreased the chance of all-cause death by 17% and improved all-cause mortality by 1.7 years compared with placebo. This effect occurred at all age ranges. One assumption of this study is that dapagliflozin use will remain consistent during the entire lifetime and that patients will always be eligible for dapagliflozin. This study also assumes that HF events do not depend on the duration of dapagliflozin treatment, but only on the age of the patient and what treatment they are on at the time they occur. Additionally, this model assumes that patients remain in the level of health they were when enrolling in the trial. A limitation of using the short-term data from the DAPA-HF trial is that it cannot truly identify time-varying treatment effects – while these were tested in a sensitivity analysis, they cannot be entirely excluded. If the effect of dapagliflozin was set to attenuate with time, these analyses would over-estimate the event-free survival time and all-cause mortality benefits. A final limitation is the relatively small sample size of the youngest subgroup of patients.
Click to read the study in JAMA Cardiology
Click to read an accompanying editorial in JAMA Cardiology
Relevant Reading: Effect of Dapagliflozin on Worsening Heart Failure and Cardiovascular Death in Patients With Heart Failure With and Without Diabetes
In-Depth [randomized controlled trial]: The DAPA-HF trial evaluated patients with HFrEF from 410 sites in 20 countries safety and efficacy outcome measures who were randomized to 10 mg dapagliflozin once daily or placebo once daily. Participants were adults with HFrEF (New York Heart Association (NYHA) classification II – IV symptoms and left-ventricular EF ≤40%, and elevated plasma N-terminal pro-B-type natriuretic peptide), who were optimally treated under local guidelines using pharmacologic and device therapy, who did not have hypotension, compromised estimated glomerular filtration rate or kidney failure, type 1 diabetes, or other life-limiting diseases. This study estimated the mean event-free survival time beyond the 17.6±5.2 months follow-up of the DAPA-HF trial. A sensitivity analysis and simulations (which varied the hazard ratio and treatment effect) were performed to check if the treatment effect changed over time. 4744 patients (76.6% men; 66.3±10.9 years) were enrolled. On average, patients experienced their HF event at age 68.3±10.9 years old or died of any cause by 69.1±10.6 years old. Dapagliflozin reduced the risk of HF events by 26% (0.74; 95% CI, 0.65-0.85) and death from any cause by 17% (0.83; 95% CI, 0.71-0.97) compared with placebo. Dapagliflozin improved event-free survival at all ages. Specifically, dapagliflozin-treated patients lived an additional 3.0 years event-free if they were 45 (95%CI, 0.1 to 6.0) or 55 years old (95% CI, 1.2 to 4.7 years; P = 0.001), 2.1 years if they were 65 years old (95%CI, 0.8 to 3.3 years; P = 0.002), 1.2 years if they were 75 years old (95%CI, 0.1 to 2.3 years; P = 0.04), and 1.6 years if they were 80 years old (95% CI, 0.5 to 2.6). These effects persisted if patients were on quadruple heart therapy (2.1 years, 95% CI, –2.5 to 6.8 years) or just dapagliflozin (1.8 years, 95%CI, 0.6 to 3.1 years). The sensitivity analysis did not demonstrate any effect of time on the effect of dapagliflozin at successfully improving HF event-free time (P = 0.26). Simulation analysis projected a similar improvement in event-free survival with dapagliflozin compared to control. If the simulation was set to attenuate the effects over time of dapagliflozin (the most conservative estimate), the event-free survival time remained at 0.7 years greater than the placebo control group. Dapagliflozin improved all-cause mortality by 1.7 years (95% CI, 0.1 to 3.3 years; P = 0.03) compared to placebo at all age ranges. The sensitivity analysis suggested that time also did not affect dapagliflozin’s effect on all-cause mortality (P = 0.95).
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.