Selpercatinib continues to demonstrate effective response in RET fusion-positive NSCLC
1. Selpercatinib was found to have similar effective objective response rates compared to past studies in both treatment-naïve and chemotherapy pretreated patient populations.
2. This study established a longer duration of response and progression-free survival in both patient populations treated with selpercatinib.
Evidence Rating Level: 2 (Good)
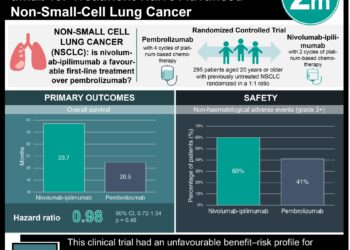
Study Rundown: Selpercatinib was approved for the treatment of RET fusion-positive NSCLCs in 2020 based on a phase I/II trial. This study provided an updated assessment of the efficacy and safety based on the original trial with additional patients and 18 more months of follow-up. The study spanned multiple countries and included both treatment-naïve as well as platinum-based chemotherapy pretreated patients. Selpercatinib was used as a daily therapy and patients had regular radiologic assessments and follow-up visits. The primary and secondary endpoints included objective response rate (ORR), progression-free survival (PFS), median duration of response (DoR), overall survival (OS), and safety. This study found ORRs that were consistent with prior data points, with a higher ORR in treatment-naïve patients compared to pretreated patients (84% vs 61%). This study also established the durability of selpercatinib therapy with DoR and PFS in treatment-naïve populations (20.2 and 22.0 months respectively) and extended the previously found DoR and PFS in pretreated patients (28.6 and 24.9 months respectively). The most common adverse events included hypertension and elevated liver enzymes. Strengths of this study included the inclusion of both types of patient populations (treatment-naïve vs pretreated), the larger sample size compared to past studies, and a longer follow-up time. Limitations to this study included a lack of a control group. Overall, selpercatinib had an effective objective response in RET fusion-positive NSCLCs and this study further supports this agent as a treatment modality.
Click to read the study in JCO
Click to read an accompanying editorial in JCO
Relevant Reading: Efficacy of Selpercatinib in RET Fusion–Positive Non–Small-Cell Lung Cancer
In-Depth [prospective cohort]: The study spanned 85 sites in 16 countries and involved patients with ECOG 0-2 with confirmed RET fusion-positive NSCLC, including those with known brain metastases. selpercatinib in doses ranging from 20mg daily to 160mg twice daily was given continuously in 28-day cycles. 356 total patients, consisting of 69 treatment-naïve and 247 chemotherapy pretreated patients were analyzed. For treatment-naïve patients, the ORR was 84% (95%CI, 72 to 92), with a 6% complete response (CR). The median DoR was 20.2 months (with a follow-up of 20.3 months), and median PFS 22.0 months (with a follow-up of 21.9 months). For chemotherapy pretreated patients, the majority received 2 or more previous lines of treatment, and had an ORR of 61% (95%CI, 55 to 67), with a 7% CR. The median DoR was 28.6 months (with a follow-up of 21.2 months), and median PFS 24.9 months (with a follow-up of 24.7 months). A subgroup of 106 patients with brain metastases at baseline showed that median intracranial PFS was 19.4 months (with a follow-up of 22.1 months) regardless of pretreatment status. Safety was assessed in 796 patients with a total exposure time of over 16,000 months and found that the most common adverse events of grade 3 or higher included hypertension (13.2%) and AST/ALT elevations (6.3%/9.0%). The most common treatment-related significant adverse event was drug hypersensitivity (1%). Overall, this study demonstrated that selpercatinib had a high ORR and established a longer DOR and PFS in patients with RET fusion-positive NSCLC.
Image: PD
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.