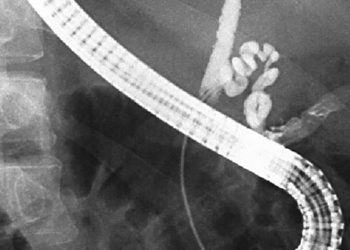
Significant morbidity among children observed following reintroduction of high-powered magnets into US market
1. This multi-center, retrospective cohort study identified nearly 600 cases of pediatric patients seeking care for high-powered magnet exposure, with about half requiring endoscopic and/or surgical intervention and nearly 10% suffering a life-threatening morbidity.
2. Ingestion of two or more magnets and developmental delay were independently associated with increased risk for morbidity, findings which contribute to the on-going discussion about these products’ safety in the consumer marketplace.
Evidence Rating Level: 2 (Good)
Study Rundown: In 2014, a Consumer Product Safety Commission injunction prevented the sale of high-powered magnet sets, based on an uptick in magnet-associated injuries after the products’ introduction in 2008. Manufacturers won the right to resume distribution in 2016, citing incomplete data regarding the injury risk these products posed to children. Researchers at twenty-five US children’s hospitals participated in a retrospective cohort study to identify children presenting for high-powered magnet exposure between January 2017 and December 2019. 596 patients ranging from 2 months to 18 years were found, with an overwhelming majority seeking care for ingestion of two or more magnets. Of this cohort, approximately half of the cases required endoscopic or surgical intervention, and nearly 10% of patients suffered intestinal perforation, fistula formation, or another significant morbidity. Increases in morbidity risk were found for patients with developmental delay, as well as ingestion of >1 magnet and magnets <5mm in diameter. Though the identification of patients presenting to tertiary centers may overestimate morbidity risk, the large patient population and convincing stratification of risk factors strengthen this first-of-its-kind database of pediatric high-powered magnet exposures. This study’s delineation of multiple facets of these cases – from the imaging-associated radiation exposure to the suspected location of magnet ingestion – provides crucial context for the on-going medico-legal debate regarding the safety of these products.
Click to read the study in PEDIATRICS
Relevant Reading: Increase in pediatric magnet-related foreign bodies requiring emergency care.
In-Depth [retrospective cohort]: Twenty-five US children’s hospitals identified charts of patients seen for foreign body ICD-10 codes between 1/1/17 and 12/31/19. Cases of high-powered magnet ingestion were included after manual review and a standardized chart abstraction process. 596 patients, aged 2 months to 18 years, were found. 50% of cases required hospitalization. 46.3% of cases required either endoscopy or surgical intervention, and 9.6% of patients suffered from some form of gastrointestinal morbidity. Risk for morbidity was increased for patients with developmental delay (OR 3.25, 95% CI 1.81-5.85), who presented for care more than once before definitive diagnosis (21.28, 6.84-66.19), and who did not spontaneously pass the magnets (40, 9.6-164). Aside from respiratory distress, all other categorized symptoms on presentation were positively associated with morbidity, with abdominal distension carrying the greatest risk. Magnets <5mm were 7-fold more likely to cause injury (p=0.001). No injury occurred with only one ingested magnet, with 2 magnets and 3+ magnets carrying a 7.5% and 18.7% risk for morbidity, respectively (p<0.001). Sex, race, and ethnicity had no bearing on morbidity risk.
Image: PD
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.