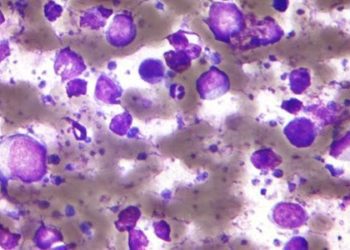
Survival increased in chronic lymphocytic leukemia patients treated with ibrutinib and rituximab
1. Previously untreated chronic lymphocytic leukemia (CLL) patients experienced increased progression-free and overall survival when treated with ibrutinib-rituximab compared to current standard chemoimmunotherapy.
2. Rates of high grade adverse events were similar between treatment groups.
Evidence Rating Level: 1 (Excellent)
Study Rundown: CLL is a common hematologic malignancy often treated with a combination of fludarabine, cyclophosphamide, and rituximab. While efficacious, there are notable side effects to the chemoimmunotherapy regimen. The Bruton’s tyrosine kinase (BTK) inhibitor ibrutinib has shown efficacy in patients with relapsed or refractory CLL, though how it performs as a first line treatment is not yet determined. This phase 3 trial evaluated ibrutinib-rituximab in comparison to standard chemoimmunotherapy in untreated CLL patients and found both progression-free and overall survival to be improved relative to standard therapy at almost 3 years after treatment initiation. The incidence of grade 3 or 4 adverse events was similar in the treatment groups, though infections were more common in the ibrutinib-rituximab group.
This randomized trial provides support for use of a new regimen as a first line treatment in CLL patients. Strengths of the study include its randomized design, mutational analysis of immunoglobulin heavy-chain variable region (IGHV) patients, and extensive subgroup analysis. The utility of this study is limited by its discordance with other trials evaluating ibrutinib in CLL.
Click to read the study in NEJM
Relevant Reading: Randomized trial of ibrutinib vs ibrutinib plus rituximab in patients with chronic lymphocytic leukemia
In-Depth [randomized controlled trial]: This phase 3, multicenter, randomized controlled trial enrolled patients between 2014 and 2016. Eligible patients were under 70 years of age and had previously untreated CLL or the small lymphocytic lymphoma (SLL) subtype of CLL. Patients with a 17p13 mutation were excluded because of known poor response to chemoimmunotherapy treatment. Patients were stratified and randomized to an ibrutinib-rituximab (n=354) or chemoimmunotherapy group receiving fludarabine, cyclophosphamide, and rituximab (n=175). At a 3 year timepoint, the primary endpoint of progression-free survival was 89.4% (95% confidence interval [CI], 86.0 to 93.0) in the ibrutinib-rituximab and 72.9% (95% CI, 65.3 to 81.3) in the chemoimmunotherapy group (hazard ratio for progression or death, 0.35; 95% CI, 0.22 to 0.56; P<0.001). Overall survival in the ibrutinib-rituximab group was 98.8% (95% CI, 97.6 to 100) compared to 91.5% (95% CI, 86.2 to 97.0) in the chemoimmunotherapy group (hazard ratio for death, 0.17; 95% CI, 0.05 to 0.54; P<0.001). In prespecified subgroup analysis ibrutinib-rituximab treatment was superior for patients regardless of age, sex, or Rai stage. An overall response occurred more in the ibrutinib-rituximab patients. Grade 3 or 4 adverse events occurred in approx. 80% of patients in both treatment groups.
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.