Trastuzumab deruxtecan superior to trastuzumab emtansine in patients with metastatic HER2 breast cancer
1. Longer progression-free survival was found in patients taking trastuzumab deruxtecan compared to trastuzumab emtansine
2. Adverse events were more common in patients taking trastuzumab deruxtecan than those taking trastuzumab emtansine
Evidence Rating Level: 1 (Excellent)
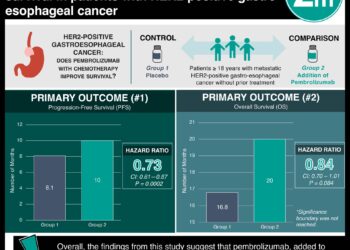
Study Rundown: This study aimed to compare the progression-free survival (PFS) of women with human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer receiving either trastuzumab emtansine (TE), the standard treatment, or trastuzumab deruxtecan (TD), a HER2 antibody-drug conjugate. Secondary outcomes included the safety profile as measured by adverse events (AEs). The PFS was 6.8 months in the TE group and unobtainable in the TD group. There were 34.1% of patients still alive in the TE group at 1 year, compared to 75.8% in the TD group, according to blinded central review. Nausea was the most commonly reported drug-related AE of any severity, followed by fatigue and vomiting. These AEs were higher in the TD group than the TE group. Neutropenia, thrombocytopenia, and leukopenia were the most common severe drug-related AEs. Neutropenia and leukopenia were higher in the TD than the TE group, while thrombocytopenia was higher in the TE than the TD group. Interstitial lung disease or pneumonitis was more common in the TD group than in the TE group and accounted for 8.2% and 1.1% of treatment discontinuations respectively. Generally, discontinuation due to AEs was more common in the TD group compared to the TE group. Limitations to this study include that some measures were not obtained by the study cutoff date, including median overall survival. Overall, trastuzumab deruxtecan appears to reduce the risk of disease progression as compared to trastuzumab emtansine.
Click to read the study in The New England Journal of Medicine
Relevant Reading: Trastuzumab deruxtecan in previously treated HER2-positive breast cancer
In-Depth [randomized controlled trial]: This multi-national, phase 3, open-label, randomized controlled trial randomly assigned 524 female patients with HER2-positive metastatic breast cancer on a 1:1 basis to receive either trastuzumab emtansine (TE) or trastuzumab deruxtecan (TD). There were 263 in the TE group and 261 in the TD group. The medications were provided by intravenous route every 3 weeks. The median PFS was 6.8 months in the TE group (95% confidence interval (CI)), while it was unobtainable in the TD group (95% CI, 18.5 months to ‘could not be estimated’). According to blinded central review, the percent of patients alive at 1 year without disease progression was 34.1% in the TE group (95% CI, 27.7% to 40.5%) compared to 75.8% (95% CI, 69.8% to 80.7%) in the TD group (hazard ratio (HR) 0.28; 95% CI, 0.22 to 0.37). Nausea, fatigue, and vomiting were the most common drug-related AEs, and were higher in the TD group than the TE group. Severe drug-related AEs included neutropenia, thrombocytopenia, and leukopenia. Neutropenia and leukopenia were higher in the TD than in the TE group (19.1% vs 3.1%, 6.6% vs 0.4%), while thrombocytopenia was higher in the TE than in the TD group (24.9% vs 7.0%). Interstitial lung disease or pneumonitis was more common in the TD group than in the TE group (10.5% vs 1.9%) and resulted in treatment discontinuation in 8.2% and 1.1% of each group, respectively. Serious AEs presented in 18.0% of TE patients, and 19.1% of TD patients. Discontinuation due to AEs was more common in the TD group (13.6%) compared to the TE group (7.3%).
Image: PD
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.