Vemurafenib plus rituximab associated with complete response in refractory hairy-cell leukemia
1. Vemurafenib plus rituximab treatment was associated with complete response for refractory or relapsed hairy-cell leukemia patients.
2. Post hoc analysis suggested minimal residual disease negative status and the lack of prior v-raf murine sarcoma viral oncogene homolog B1 (BRAF) inhibitors exposure were associated with better survival.
Evidence Rating Level: 2 (Good)
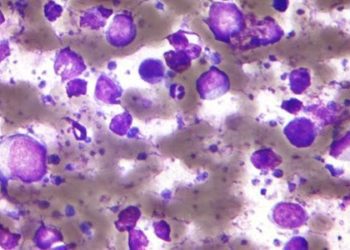
Study Rundown: Hair-cell leukemia (HCL) is a B-cell neoplasm that is driven by the BRAF V600E activating mutation. BRAF V600E inhibitors have been shown, in previous studies, to be an encouraging treatment, but residual disease remained after monotherapy treatment. The resistant cancer cells still maintain their CD20 receptors, highlighting the potential for a dual blockade. As such, this study treated relapsed or refractory hairy-cell leukemia patients with a combination of vemurafenib, a BRAF inhibitor, and rituximab, and anti-CD20 monoclonal antibody. The study determined most patients had a complete response with no minimal residual disease after treatment. The adverse event profile was consistent with previous studies of monotherapies. The adverse events consisted of infusion reactions for rituximab along with photosensitivity, arthralgias, and cutaneous rashes for vemurafenib. The study was limited by the small sample size and lack of a comparator arm. Nonetheless, the study’s results are significant, as the trial demonstrated a short, chemotherapy-free, nonmyelotoxic regimen of vemurafenib plus rituximab was associated with a complete response in refractory or relapsed hairy-cell leukemia.
Click here to read the study in the NEJM
Relevant Reading: BRAF Mutations in Hairy-Cell Leukemia
In-Depth [prospective cohort]: The single-arm, single-center, phase 2 clinical trial enrolled 31 patients in Italy. The study inclusion criteria consisted of patients with refractory or relapsed HCL, mutated BRAF V600E, and previous treatment with a BRAF inhibitor. Patients without cytopenia were excluded from the study. All patients were treated with eight weeks of vemurafenib (960 mg, twice daily) and eight rituximab infusions (375 mg per square meter of body-surface area) over an 18 week period. The primary endpoint was a complete response at the end of planned treatment, which was assessed four weeks after the last rituximab dose. A complete response was defined as resolution of cytopenias, no palpable splenomegaly, and no hairy cells visible in bone marrow biopsy or blood smear samples. Minimal residual disease (MRD) was assessed by polymerase-chain-reaction (PCR) for BRAF V600E in bone-marrow and peripheral samples. At the end of the treatment period, 87% (26 out of 30) of patients had a complete response, out of which, 65% (17 out of 26) had minimal residual disease resolution. Progression-free survival from the intention-to-treat population was 78% at a median follow-up of 37 months. Drug-related adverse events included infusion-related reactions, transient neutropenia, rash, and photosensitivity. Post hoc analysis suggested that MRD negativity and no prior exposure to BRAF inhibitors correlated to improved survival. In general, the study found a vemurafenib plus rituximab treatment was associated with complete response in refractory or relapsed hairy-cell leukemia patients.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.