#VisualAbstract: All-oral regimen with delamanid, linezolid, levofloxacin, and pyrazinamide is effective for fluoroquinolone-sensitive multidrug-resistant tuberculosis
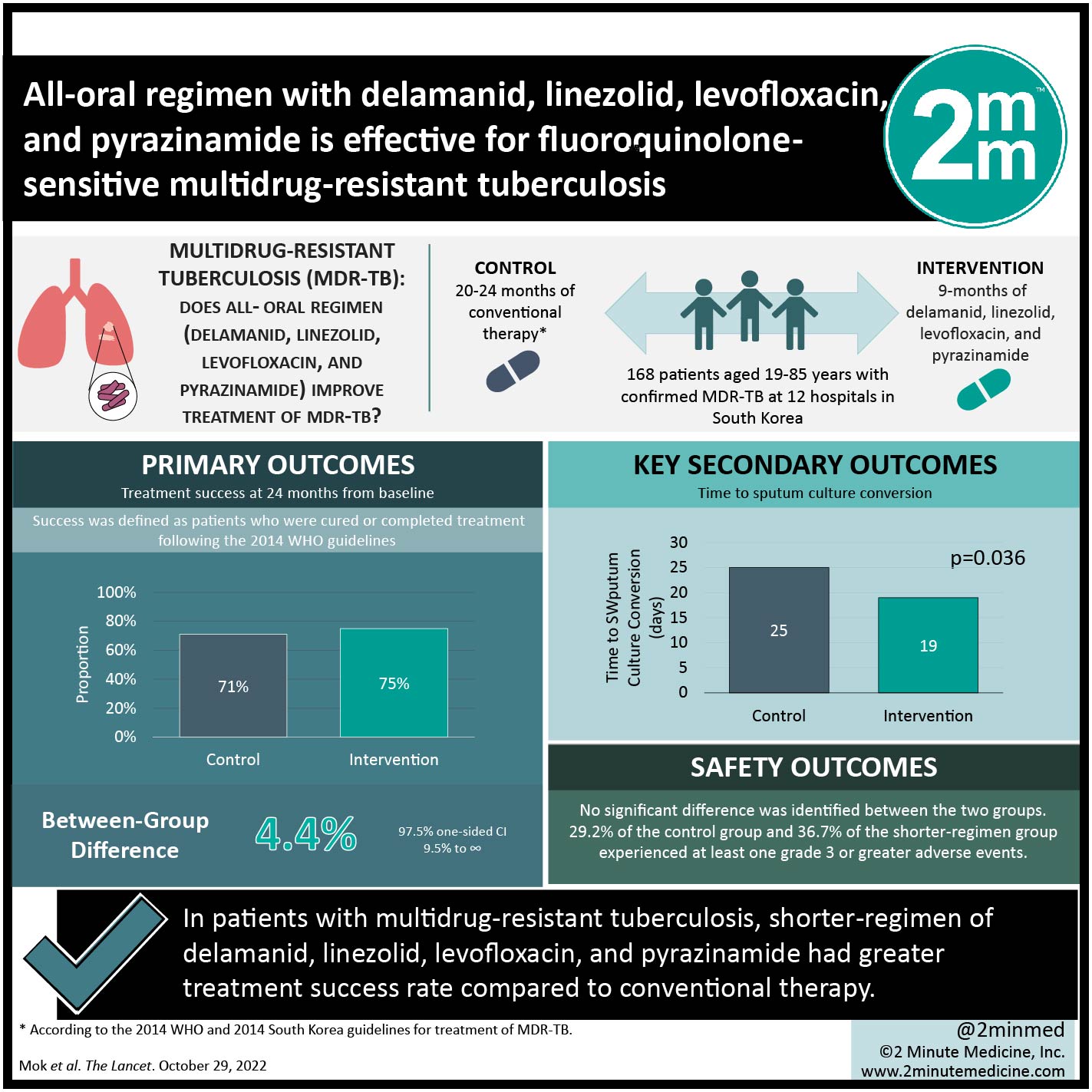
1. At 24 months, treatment success rate was numerically greater in the shorter-regimen group versus conventional therapy, satisfying the noninferiority criteria.
2. Time to culture conversion in solid media was quicker in the intervention group compared to conventional therapy.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Multidrug-resistant tuberculosis (MDR-TB) is an infection by Mycobacterium tuberculosis that is resistant to isoniazid and rifampicin. Conventional therapy for MDR-TB includes at least 20 months of anti-TB drugs with an injectable for 8 months. However, given the extensive treatment period, adherence to the treatment regimen can be variable. New all-oral drug regimens have been developed that may shorten treatment duration while having comparable efficacy. This randomized trial aimed to investigate the safety and efficacy of all-oral regimen (delamanid, linezolid, levofloxacin, and pyrazinamide) versus conventional therapy for treatment of fluoroquinolone-sensitive multidrug-resistant tuberculosis. The primary outcome was treatment success at 24 months while key secondary outcome included time to sputum culture conversion. According to study results, the shorter, all-oral treatment group was noninferior to conventional therapy after 24-months of follow-up. This study was strengthened by a large sample size with individuals of all ages, thus increasing its credibility.
Click to read the study in The Lancet
Relevant Reading: Four-Month Rifapentine Regimens with or without Moxifloxacin for Tuberculosis
In-depth [randomized-controlled trial]: Between Mar 4, 2016, and Sept 14, 2019, 443 patients were screened for eligibility at 12 hospitals in South Korea. Included were those aged 19-85 years with confirmed multidrug-resistant tuberculosis. The intervention group received 9-months of delamanid, linezolid, levofloxacin, and pyrazinamide, while the conventional therapy group received 20-24 months of treatment according to the 2014 WHO and 2014 South Korea guidelines for treatment of MDR-TB. Altogether, 168 patients (89 in control group and 79 in shorter-regimen group) were included in the intention-to-treat analysis. Median age of patients was 47 years, and the majority were men (69%). The primary outcome of treatment success at 24 months from baseline was numerically greater in the intervention group (54 of 72, 75%) than in the control group (60 of 85, 71%; between-group difference 4.4% [97.5% one-sided CI 9.5% to ∞, satisfying the non-inferiority criteria). In solid media, the time to culture conversion was significantly shorter in the 9-month treatment group compared to the conventional 20–24-month treatment group (19 days vs. 25 days, respectively; p=0.036). Overall, findings from this study suggest that 9-month treatment with delamanid, linezolid, levofloxacin, and pyrazinamide may be effective in patients with fluoroquinolone-sensitive multidrug-resistant TB.
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.