Additional aromatase inhibitor may improve endometrial cancer chemotherapy effectiveness
1. In a phase II trial of 35 women with advanced endometrial cancer, combination therapy with aromatase inhibitor (letrozole) and mTOR inhibitor (everolimus) demonstrated a high clinical benefit rate.
Evidence Rating Level: 2 (Good)
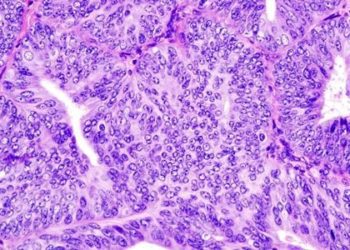
Study Rundown: Endometrial cancer (EC) is the most common gynecological malignancy in the United States with over 52 000 cases diagnosed in 2014. Previous studies have noted an association between dysregulation of the phosphoinositol-3 kinase (PI3K)/mammalian target of rapamycin (mTOR) pathway with poorer prognosis in EC patients. Estrogen receptor (ER) signaling have been shown to cross-regulate the PI3K/mTOR signaling pathway and may provide a potential target for mTOR signaling inhibition. The purpose of this phase II clinical trial was to evaluate the response of a combination therapy of an aromatase inhibitor (letrozole) and PI3K/mTOR inhibitor (everolimus) in patients with recurrent EC. Overall, 35 patients with recurrent/metastatic EC were treated with the combination therapy. At the conclusion of the trial, the combination treatment demonstrated a clinical response rate of 40% at 16 weeks, as measured by prolonged stable disease. The safety profile of this combination was excellent as there were no patients that discontinued treatment due to toxicity. The most significant predictor of response were patients with endometrioid histology. The results of this trial support the use of everolimus and letrozole in women with advanced endometrioid EC. However, this was only a phase II trial with a small sample size; additional larger randomized controlled trials are required to accurately describe the efficacy of this combination treatment.
Click to read the study in JCO
Relevant Reading: A phase 2 study of the oral mammalian target of rapamycin inhibitor, everolimus, in patients with recurrent endometrial carcinoma.
In-Depth [prospective cohort]: This clinical trial was a phase II, open-labelled, multi-centered trial to determine the effectiveness of the combination therapy of letrozole and everolimus in patients with advanced EC. Overall, 35 patients (median age 62 years; range: 24-82 years) with progressive or recurrent EC were recruited to receive daily doses of oral letrozole and everolimus for 4 week cycles. The primary end point was clinical benefit rate (CBR), defined as complete response, partial response, or stable disease for 16 weeks or more. After a median follow-up of 14 months, the CBR was 40% (14 patients) at 16 weeks with a median number of cycles of 15 (range: 7-29 cycles). Twenty percent of patients discontinued treatment after prolonged complete response. The most common side-effects included fatigue (74%), hypercholesterolemia (71%), and mucositis (66%); no patients discontinued treatment due to adverse events. The median overall survival time was 14 months (95% CI: 9.5 to 24.4 months). Patients with endometrioid histology demonstrated significantly increased CBR compared to patients with serous histology (37.5% vs 0%; p<0.0092).
More from this author: Rituximab linked with reduced chronic immune disease following stem cell transplantation, High-dose prophylaxis for hemophilia increases costs with minimal benefit, Ambrisentan found ineffective against idiopathic pulmonary fibrosis
Image: PD
©2014 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. No article should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2 Minute Medicine, Inc.




![Vaccines for predicted influenza strains may provide wide protection [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2014/11/18156_lores-75x75.jpg)
