Combined Encorafenib-Binimetinib-Cetuximab therapy associated with longer overall survival in patients with BRAF V600E-mutated coloreactal cancer
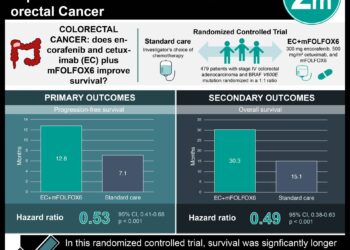
1. A phase 3 randomized clinical trial showed that combined therapy using a BRAF inhibitor (encorafenib), EGFR monoclonal antibody (cetuximab) and a MEK inhibitor (binimetinib) led to longer overall disease survival in patients with BRAF V600E mutated metastatic colorectal cancer as compared to control groups who received a single agent.
2. Doublet therapy with encorafenib-cetuximab was also shown to have longer overall disease survival as compared to control.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Colon cancer patients with the BRAF V600E mutation have been shown to have poorer prognoses. Approximately 10% of patients with metastatic colon cancer have this mutation, and standard chemotherapy regimens have shown to have limited success. A novel BRAF inhibitor, encorafenib, was studied in this trial in conjunction with EGFR monoclonal antibody treatment (cetuximab) and MEK inhibitor treatment (binimetinib) to evaluate whether combined treatment would result in longer disease survival as compared to the standard therapy available to patients with BRAF V600E mutated colorectal cancer. Overall, researchers found that combined triple therapy led to longer overall survival as compared to control. They also found that combined therapy with two experimental agents also led to longer overall survival as compared to control. These results are promising in creating new novel agents for treating metastatic colorectal cancer in this particular subset of patients, however more research is warranted so that further differences in treatment outcomes between triplet therapy and doublet therapy can be investigated.
Click here to read the study in NEJM
In-Depth [randomized clinical trial]: This open-label, phase 3 clinic trial randomized 665 patients with BRAF V600E mutation into one of three treatment groups in a 1:1:1 ratio: triplet-therapy with encorafenib-binimetinib-cetuximab (n=244), doublet-therapy with encorafenib-cetuximab (n=220) or control with either cetucimab-irinotecan, cetuximab-folfiri or fluorouracil-irinotecan (n=221). All treatment was administered in 28-day cycles until disease progression, toxic effects, death or withdrawal from the study occurred. The primary endpoint of this study was to compare overall survival in the triplet-therapy group as compared to the control group. Secondary endpoints included overall survival in the doublet-therapy group verses the control group, and progression-free survival, duration of responses, and safety profiles of all three arms. The median time to follow up for all three groups was 7.8 months. Median overall survival was 9.0 months in the triplet-therapy group as compared to 5.4 months in the control group (95% CI 4.8-6.6). The risk of death was noted to be significantly lower in the triplet-therapy group compared to control (48% lower risk; HR 0.52, 95% CI 0.39 to 0.70, p <0.0001). Similarly, the mean overall survival was also longer in the doublet-therapy group compared to control (8.4 months vs 5.4 months; 95% CI 7.5-11.0) and the risk of death was also significantly lower in the double-therapy group (HR 0.60; 95% CI 0.45-0.79; p<0.001). Progression-free survival was longer in the triplet and doublet therapy groups compared to control (4.3 months versus 4.2 months verses 1.5 months respectively; HR 0.38, 95% CI 0.29 to 0.49 for triplet versus control; HR 0.40, 95% CI 0.31 to 0.52 for doublet versus control). Grade 3 or higher adverse events were noted in 58% of patients in the triplet group, 50% of patients in the doublet group and 61% of patients in the control group. The most common adverse events included diarrhea, nausea, vomiting and acneiform dermatitis.
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.