Decompressive craniectomy linked with decreased mortality in TBI: The RESCUEicp trial
1. Patients with traumatic brain injury (TBI) and refractory intracranial hypertension following 2 stages of standard therapy who were then randomized to receive decompressive craniectomy had lower rates of mortality and higher rates of vegetative state at 6 months compared to those treated with standard medical therapy alone.
2. Adverse events occurred significantly more frequently in the surgical group, though survivors in the surgical group had significantly shortened stays in the ICU.
Evidence Rating Level: 1 (Excellent)
Study Rundown: TBI can be complicated by elevated intracranial pressure (ICP) secondary to hematomas, contusions, diffuse brain swelling, or hydrocephalus. The role of decompressive craniectomy, in which a large section of the skull is removed to relieve increased pressure and then reconstructed during a follow-up interval, remains unclear. A prior trial deploying craniectomy for patients with elevated ICP after first-tier approaches were utilized revealed more unfavorable outcomes in the craniectomy group compared to the medically treated group. In the Randomized Evaluation of Surgery with Craniectomy for Uncontrollable Elevation of Intracranial Pressure (RESCUEicp) trial, decompressive craniectomy was deployed as as last-tier intervention.
Patients with uncontrollable intracranial hypertension following two tiers of standard therapy were randomized to receive expedited craniectomy, while those in the medical group continued to receive stage 1 and 2 treatments (including basic measures, such as head elevations, ventilation, sedation, and medical therapies such as use of hypertonic solution). The authors found that patients in the craniectomy group had significantly decreased mortality at 6 and 12 months as well as increased rates of vegetative state; the outcome of an upper severe disability or better was higher in the craniectomy group. The results of this trial will help inform patients and clinicians in balancing the potential benefits of decompressive craniectomy for TBI with the risk of a vegetative state as an outcome. The results of the trial were limited by the lack of reporting of outcomes related to cranial reconstruction, which generally took place a few weeks to months after the craniectomy.
Click to read the study, published in NEJM
Relevant Reading: Decompressive craniectomy in diffuse traumatic brain injury
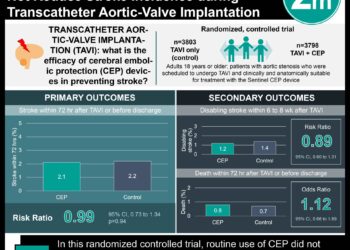
In-Depth [randomized controlled trial]: In this international, multicenter trial, 408 patients with TBI and refractory intracranial hypertension (defined as >25 mmHg) were randomized either to receive decompressive craniectomy or continued medical therapy. This intervention was offered as a last-tier, following standard medical therapies. Patients that were selected for surgery either underwent large unilateral frontotemporoparietal or bifrontal craniectomy, with the exact procedure left up to the discretion of the surgery. The primary outcome measure was the Extended Glasgow Outcome Scale (GOS-E) at 6 months following randomization. This scale scores individuals from death and vegetative state on one end of the spectrum to “upper good recovery” on the other, with 8 stages that reflect varying levels of dependent versus independent function. The primary outcome differed significantly between the groups (p < 0.001), allowing the authors to report descriptive statistics of the GOS-E outcomes: death, 26.9% in the surgical group vs 48.9% in the medical group; vegetative state, 8.5% vs 2.1%; lower severe disability (dependent on others for care), 21.9% vs 14.4%; upper severe disability (independent at home), 15.4% vs 8.0%; moderate disability, 23.4% vs 19.7%; good recovery, 4.0% vs 6.9%. Adverse events were more common in the craniectomy group (16.3% vs 9.2%, p = 0.03), though patients in the surgical group who survived had a shorter median time in the ICU.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.