Endoscopic screening for esophageal cancer may reduce long-term mortality
1. In a prospective cohort trial of over 3000 volunteers who live in areas with high incidence of esophageal cancer, one-time screening with endoscopy was associated with increased detection of pre-neoplastic lesions and reduced long-term mortality compared to control.
Evidence Rating Level: 3 (Average)
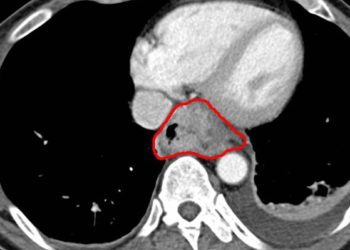
Study Rundown: Esophageal squamous cell carcinoma (ESCC) is the eighth most common cancer worldwide and carries an extremely poor prognosis, in large part due to the advanced stage typically found at diagnosis. Previous studies have investigated various screening methods in high risk population groups and have demonstrated that endoscopic exam with Lugol’s iodine staining and biopsy has high sensitivity and specificity for detecting precancerous lesions. The purpose of this study was to evaluate the long-term cancer incidence and mortality among a cohort of residents in rural China who had undergone one-time endoscopic screening and treatment of precancerous lesions. Researchers prospectively screened over 3000 volunteers with one-time endoscopy and was compared to over 700 volunteers in the control group without screening. At the conclusion of the trial, the screening cohort had significantly lower incidence of ESCC, lower ESCC-related mortality, and lower total cancer incidence and mortality compared to the control arm at 10 years follow-up. This was the first large, population-based study to evaluate the effect of endoscopic screening on long-term ESCC incidence and the results of this trial support the use of this modality in regions with high-incidence of ESCC. However, there were limitations in the selection of the intervention and control groups, resulting in significant differences between the two groups, which may confound the protective effect of endoscopy. Furthermore, an effective screening program requires the availability of highly skilled endoscopists, which may limit the applicability to resource-limited areas. Future evaluation of large-scale endoscopy screening programs are required to confirm this effect.
Click to read the study in JCO
Relevant Reading: Early detection of superficial squamous cell carcinoma in the head and neck region and esophagus by narrow band imaging: a multicenter randomized controlled trial
In-Depth [prospective cohort]: This prospective trial involved the recruitment of volunteers from 14 villages in Northern China with high population rates of ESCC. Another 10 villages south of the area were selected as control communities. Eligible patients were age 40 to 69 years with no contraindication to endoscopy. The primary end points were incidence of ESCC and ESCC-related mortality, and secondary end points included overall cancer incidence and mortality. A total of 3319 subjects underwent one time endoscopy with Lugol’s iodine staining and biopsy of suspicious lesions. Abnormal biopsy findings were treated by clinical guidelines with endoscopic follow-up at 6 and 12 months. At 10 years follow-up, ESCC was seen in 4.17% and 5.93% of the screening and control groups, respectively (p<0.001). ESCC mortality was 3.35% and 5.05% in the screening and control arms, respectively (p < 0.001). Total cancer incidence (p = 0.02) and total cancer mortality (p < 0.001) were lower in the intervention group compared to the control group.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.