Frozen fecal microbiota transplantation effective for recurrent Clostridium difficile
1. Using frozen versus fresh fecal microbiota transplants for patients with recurrent or refractory Clostridium difficile infection resulted in similar rates of resolution of diarrhea at 13 weeks of follow-up.
2. Both frozen and fresh fecal microbiota transplantation had similar rates of adverse events after 13 months of follow-up.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Clostridium difficile infection (CDI) is associated with great morbidity in the healthcare setting and in the community. Patients can experience prolonged diarrhea and even require surgical intervention for its complications. In fact, more than 60% of patients have recurrent episodes that are particularly resistant to treatment. Increasing numbers of studies demonstrate that fecal microbiota transplantation (FMT) restores protective colonic bacteria and is effective for recurrent CDI. However, logistical issues make it difficult to have fresh material on hand for all patients. Thus, the purpose of this study was to determine whether frozen FMT was equivalent in efficacy and safety to fresh FMT for patients with recurrent or refractory CDI. In a randomized and double-blind trial of over 230 adults, using frozen FMT did not result in any significant difference in the rate of resolution of diarrhea at 13 weeks. Additionally, no differences existed in the rate of adverse or serious side effects between the groups.
Overall, this study suggests that for patients with recurrent or refractory CDI, frozen FMT is equally effective as fresh FMT in causing clinical resolution of diarrhea. Using frozen instead of fresh FMT may enable cheaper production and more widespread adoption of the procedure. However, the 13-week period of follow-up is likely not long enough to thoroughly evaluate for long-term adverse events. Longer follow-up is necessary to determine how FMT affects overall health.
Click to read the study, published today in JAMA
Relevant Reading: Duodenal infusion of donor feces for recurrent Clostridium difficile
In-Depth [randomized controlled trial]: This study enrolled 232 adults with recurrent or refractory CDI at six medical centers in Canada between 2012 and 2014. In a randomized, double-blind, noninferiority trial, 118 patients were assigned to receive fresh FMT and 114 patients to frozen FMT, both delivered by enema. Patients who received up to 2 FMTs, did not require antibiotics between the two FMTs for CDI, and did not require systemic antibiotics for intercurrent infection were assessed as part of a per-protocol population. Of 178 patients in the this population, 83.5% of those who received frozen FMT experienced clinical resolution of diarrhea compared to 85.1% of patients who received fresh FMT (difference -1.6%; 95%CI -10.5% to ∞; p = 0.01 for noninferiority). A modified intention-to-treat (mITT) population was also studied, which consisted of all patients receiving at least 1 FMT but who required antibiotics, received a FMT different from their first FMT, or who were lost to follow-up. Of 219 patients in the mITT population, 75% of patients receiving frozen FMT experienced clinical resolution of diarrhea compared to 70.3% of patients receiving fresh FMT (difference 4.7%; 95%CI -5.2% to ∞; p < 0.001 for noninferiority). Because the lower confidence limits in these two groups were above the noninferiority margin of -15%, frozen FMT was determined to be noninferior to fresh FMT in both populations. Side effects from FMT included mild to moderate diarrhea, nausea, and cramps. Deaths occurring during the follow-up period were not attributed to FMT, and no differences existed in the rate of side effects between frozen and fresh FMT.
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.

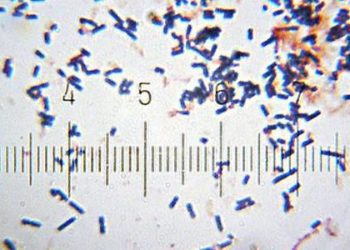
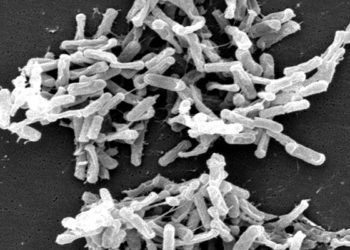
![[Physician Comment] The extent of C. difficile infections may not differ in light of immune status](https://www.2minutemedicine.com/wp-content/uploads/2012/11/x800px-Clostridium_difficile_01-e1353335581537.jpg.pagespeed.ic_.We5DxZl6Ye-350x250.jpg)