Lariat device for left atrial appendage exclusion associated with adverse events
1. In this systematic review, the off-label use of the Lariat device for left atrial appendage exclusion in patients with atrial fibrillation was associated with roughly 90% procedural success.
2. Out of 309 patients studied, there were 35 unique adverse events identified, including pericardial effusion, death, and need for urgent cardiac surgery.
Evidence Rating Level: 1 (Excellent)
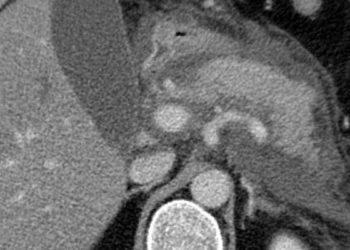
Study Rundown: Atrial fibrillation (AF) is a significant cause of cardiovascular morbidity and mortality in the United States, leading to increased risk of embolic stroke. Traditionally, anticoagulation has been one of the mainstays of therapy for stroke prevention in AF, however, 90% of thrombi causing strokes in AF originate from the left atrial appendage (LAA). This study examined the off-label use of the Lariat snare device in transcatheter LAA exclusion in patients with AF. Although the Lariat device has received FDA clearance for soft tissue approximation, the safety and effectiveness of this technique for LAA exclusion has not been well characterized.
The study found that this method was associated with about 90% procedural success. However, out of the 309 patients who were included in this systematic review, there were 35 unique adverse events, which included pericardial effusion, death, and need for urgent cardiac surgery. Strengths of this study include the systematic review methodology, which was able to identify 5 reports of Lariat use in human patients. Limitations include the poor quality of the studies that were included, as they were uncontrolled and came from a limited number of centers.
Click to read the study, published today in JAMA Internal Medicine
Click to read an invited commentary in JAMA Internal Medicine
Relevant Reading: Percutaneous Left Atrial Appendage Suture Ligation Using the LARIAT Device in Patients With Atrial Fibrillation: Initial Clinical Experience
In-Depth [systematic review]: This systematic review searched MEDLINE, EMBASE, CINAHL, and the Cochrane Library databases, using various search terms related to the Lariat device. Adverse events were identified by searching the FDA MAUDE database for events related to the Lariat device. A total of 5 reports including 309 participants were identified. The primary endpoint was successful closure of the LAA using the Lariat device. Safety endpoints included cardiac perforation, pericardial effusion, and urgent cardiothoracic surgery, among many others.
Successful closure of the LAA was achieved in 90.3% of patients. Searching the MAUDE database identified 35 unique reports of adverse events, including 5 reports noting pericardial effusion and in-hospital death, 22 reports of pericardial effusion requiring urgent cardiac surgery, and 1 report of need for urgent cardiac surgery without pericardial effusion. The FindWIRZ magnetic wires used for deployment were associated with high rates of complications, as was the Lariat device/suture tightener, with these two components accounting for 94.3% of identified complications.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.