Pediatric palliative care outcome measures often miss quality of life
1. A systematic review of outcome measures found that only a minority addressed quality of life and patient (child) reported outcomes, and many did not measure psychometric properties.
2. Commonly measured outcomes included health-related quality of life, spiritual well-being, satisfaction with care and/or communication, perceived social support, and family involvement in treatment.
Evidence Rating Level: 2 (Good)
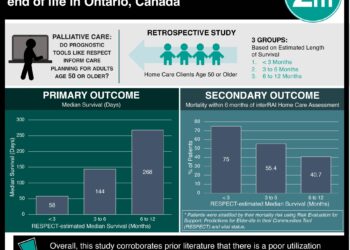
Study Rundown: Measuring outcomes in pediatric palliative care is complicated by the wide range of diseases and contexts in which palliative care is practiced. Previous research has shown that general quality of life measurements are not valid in a pediatric palliative care context, and in many cases, proxy measurements must be used depending on the nature of the disease and child age. In this systematic review, researchers reviewed literature from multiple databases to identify instruments used and outcomes measured in pediatric palliative care research between 2006 and 2016. They identified 19 studies that used 23 instruments, of which only 5 included patient-reported outcomes and only 15 included psychometric properties. Less than one-third of the 19 studies addressed quality of life, defined as “having fun or experiencing events that added meaning to life” as an outcome measure. Commonly measured outcomes included health-related quality of life, spiritual well-being, satisfaction with care and/or communication, perceived social support, and family involvement in treatment.
These findings are limited because the researchers did not have access to information regarding development and rationale of the measurement scales. Nonetheless, the study is strengthened by its broad literature review that provides the first description of the range of instruments used to assess the impact of pediatric palliative care interventions. For physicians, these findings guide future research and pediatric palliative care interventions by suggesting that children are not systematically involved in reporting outcomes, and that quality of life measurements are lacking or underutilized.
Click to read the study, published today in Pediatrics
Relevant reading: Paediatric Palliative Care Improves Patient Outcomes and Reduces Healthcare Costs: Evaluation of a Home-Based Program
In-Depth [systematic review]: Researchers conducted a primary systematic literature review of 5 databases (Embase, Scopus, The Cochrane Library, PsychInfo, and PubMed) using pediatric palliative care keywords to identify 2150 studies published from January 1, 2006, to June 1, 2016. Of these, 19 studies using 23 reported outcome instruments met inclusion criteria of having a study population aged 0 to 18 years with life-limiting diseases receiving palliative care interventions. Studies that included sickle cell disease, chronic pain, perinatal death, patients aged >18 years, economic analyses, and studies using pain as a single outcome were excluded. Of the 19 studies included, 9 were retrospective, 6 were prospective, and 4 were randomized controlled trials.
In the 19 studies, 23 different instruments were identified including the Pediatric Quality of Life Inventory 4.0, Survey About Caring for Children With Cancer, and Hospital Anxiety and Depression Scale. Instruments commonly measured health-related quality of life, spiritual well-being, satisfaction with care and/or communication, perceived social support, and family involvement in treatment. Of the 23 instruments, 5 included patient-reported outcome measures and 15 included psychometric properties. Quality of life, defined as “having fun or experiencing events that added meaning to life” was measured in 6 of the 19 studies.
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.