Pimecrolimus effective first-line treatment for atopic dermatitis
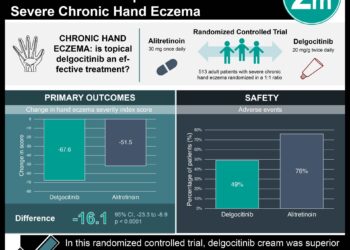
1. The majority of patients using pimecrolimus cream (PIM, 52.6%) or topical corticosteroids (TCS, 50.5%) for the treatment of mild to moderate atopic dermatitis were cleared of manifestations of the disease by week 3 of use.
2. No significant differences in adverse effects and safety or changes in immunity were seen between the PIM and TCS treatment groups.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Atopic dermatitis (eczema) is a chronic, inflammatory skin disease that affects up to 25% of all infants. TCS are typically used as first-line treatment, but many patients and parents are non-compliant due to fear of their potential side effects. Researchers of the current study sought to compare the safety and efficacy of TCS and PIM in the treatment of mild to moderate atopic dermatitis. PIM is a topical calcineurin inhibitor used as first line therapy for atopic dermatitis in sensitive skin areas. Results showed that a majority of patients in both groups were cleared of the disease by week 3 of treatment. Patients in the PIM group required a much smaller amount of subsequent steroid use for exacerbations and achieved similar results. No significant differences in adverse effects or impairment of humoral or cellular immunity were seen between groups. This study is limited by adverse effect analyses that were not adjusted for the multiplicity of comparisons. Nonetheless, these findings may influence pediatricians to use PIM as a first-line agent in the treatment of atopic dermatitis in patients whose parents may be hesitant to use TCS.
Click to read the study, published today in Pediatrics
Relevant Reading: Atopic dermatitis
In-Depth [randomized controlled trial]: A total of 2439 children, ages 3 to 12 months, with atopic dermatitis were randomized into low potency TCS or PIM treatment arms. Parents were instructed to use the allotted treatment cream until atopic dermatitis had cleared. The medication could be restarted if the disease returned, and the PIM group was told to start a TCS if an exacerbation was not controlled by the assigned treatment. Treatment success was defined as being clear or almost clear of the disease, and success was achieved at week 3 by 52.6% and 50.5% of patients in the PIM and TCS groups, respectively. At the end of the 5-year study period, 88.7% of the PIM group and 92.3% of the TCS group reached overall treatment success. Patients in the PIM group used TCSs for a median of 7 days vs. 178 days in the TCS group. The PIM group experienced significantly more episodes of bronchitis (p = .02), infected eczema (p < .001), impetigo (p = .045), and nasopharyngitis (p = .04). Similar increases in immunoglobulin levels and decreases in peripheral blood T and B lymphocytes at baseline to week 260 were seen in both groups and were considered normal when compared to historical controls.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.