Quick Take: Short-term androgen suppression and radiotherapy versus intermediate-term androgen suppression and radiotherapy, with or without zoledronic acid, in men with locally advanced prostate cancer (TROG 03.04 RADAR): 10-year results from a randomised, phase 3, factorial trial
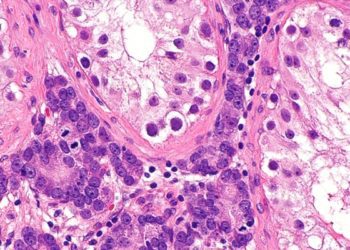
The optimal duration of androgen suppression therapy with radiotherapy for locally advanced prostate cancer is unknown. In 2018, the French Canadian Prostate Cancer Study (PCS) IV trial showed that 18 months of androgen suppression and radiotherapy is associated with fewer adverse effects than 36 months of the same regimen, suggesting that an even shorten regimen may be safer but still effective. In this randomized, controlled phase 3 trial, 1,071 men with locally advanced prostate cancer at 23 treatment centers across Australia and New Zealand were assigned in a 1:1:1:1 ratio to one of four treatment groups: 6 months of neoadjuvant androgen suppression with leuprorelin and radiotherapy (6AS+RT, control group), an additional 12 months of androgen suppression (18AS+RT), 6AS+RT with 18 months of zoledronic acid, or 18AS+RT with 18 months of zoledronic acid. The primary outcome was prostate cancer-specific mortality, and results were analyzed at 10 years. Omnibus testing for interactions at 10 years revealed that there were no significant differences between androgen suppression and zoledronic acid, and as such, the treatment groups were subsequently collapsed according to duration of androgen suppression and zoledronic acid use. Researchers found that prostate cancer-specific mortality was higher in the 6AS+RT group than in the 18AS+RT group (13.3% vs. 9.7%, respectively, sub-hazard HR (sHR) 0.70, 95% CI 0.50 to 0.98, p=0.035). However, there was no difference in prostate cancer-specific mortality between the no zoledronic acid and zoledronic acid groups (11.7% vs. 11.2%, respectively, sHR 0.95, 95% CI 0.69 to 1.32, p=0.78). There were twelve serious drug-related adverse events. A major limitation of this study was the change in the primary endpoint in 2011 from prostate-specific antigen (PSA) progression to prostate cancer-specific mortality, which compromised the power of the study. Overall, this study suggests that 18 months of androgen suppression plus radiotherapy is safe and more effective than 6 months of the same treatment regimen for locally advanced prostate cancer, but that the addition of zoledronic acid is not beneficial.
Click to read the study in Lancet Oncology
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.

![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-350x250.png)