Replacement of infusion sets every seven days may be safe and cost-effective to prevent life-threatening catheter-related bloodstream infections
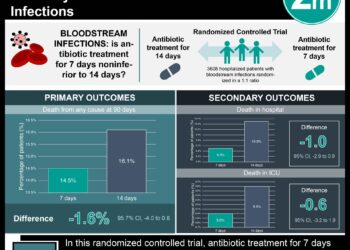
1. Prevalence of catheter-related bloodstream infections (CRBSI) was similar in the intervention (7-day) and control (4-day) groups for central venous access devices (1.78% vs. 1.46%).
2. Compared to the 4-day group, the 7-day group had mean cost savings of AU$483 for central venous access devices and AU$43 for peripheral arterial catheters.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Catheter-related bloodstream infection (CRBSI) accounts for 70% of all healthcare-related bloodstream infections. While studies suggest the optimal time interval for infusion set replacement to be four days, such a duration can have both financial and logistical limitations. This randomized controlled trial aimed to compare the effectiveness and costs of 7-day (intervention) versus 4-day (control) infusion set replacement to prevent life-threatening CRBSI in patients with central venous access devices and peripheral arterial catheters. The primary outcome was CRBSI, defined as bacteremia or fungemia and at least one positive blood culture plus matching organism(s) found on the catheter tip. Secondary outcomes included catheter tip colonization (>15 CFUs), infusion set colonization (≥103 CFU/mL) and all-cause bloodstream infection. According to study results, the proportion of patients with CRBSI in the 7-day group and 4-day group was similar, with no treatment-related adverse events. 7-day replacement was also associated with reduced consumable costs and staff time spent per patient. This study was limited by a lack of blinding between patients and clinical staff which may have introduced bias in the results. Nonetheless, this study provided valuable insight into 7-day versus 4-day infusion set replacement for the prevention of CRBSI.
Click to read the study in The Lancet
Relevant Reading: Comparison of Rates of Central Line–Associated Bloodstream Infections in Patients With 1 vs 2 Central Venous Catheters
In-depth [randomized controlled trial]: Between May 30, 2011, and Dec 9, 2016, 6007 patients were assessed across 10 Australian hospitals. Included patients were of any age, with a central venous access device or peripheral arterial catheter for ≥ 24 hours and infusion sets attached. Those with a bloodstream infection in the past 48 hours or with infusion sets replaced were excluded. Altogether, 2944 patients (1463 in the 7-day group and 1481 in the 4-day group) were included in the modified intention-to-treat analysis.
The primary outcome of CRBSI was observed in 20 (1.78%) patients in the 7-day group and 16 (1.46%) in the 4-day group for central venous access devices (CVADs, absolute risk difference [ARD] 0.32%, 95% confidence interval [CI] -0.73 to 1.37). Findings between both groups were similar for CRBSI per 1000 days (1.36 for 7-day group vs. 1.08 for 4-day group, hazard ratio [HR] 1.33, 95% CI 0.69 to 2.57, p=0.40) and time to CRBSI. For peripheral arterial catheters (PACs), 1 (0.28%) patient in the 7-day group and 0 in the 4-day group had a CRBSI (ARD 0.28%, 95% CI -0.27 to 0.83). The secondary outcomes of catheter tip colonization, mortality, all-cause bloodstream infection, and central line-associated bloodstream infection showed no significant difference for CVADs and PACs. Furthermore, relative to the 4-day group, the 7-day group had mean cost savings of AU$483 (CVAD) and AU$43 (PAC). Similarly, the median nursing time saved with 7-day infusion set replacement was 174 min (CVAD) and 7 min (PAC). There were no treatment-related adverse events. Overall, findings from this study suggest that infusion set use can be safely extended to 7 days with resultant cost and workload reductions.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.