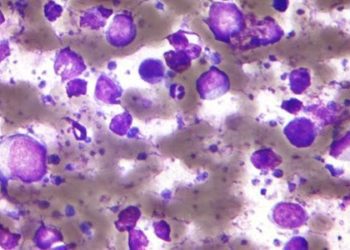
Rituximab improves survival in B-cell lineage acute lymphoblastic leukemia
1. Compared to standard chemotherapy alone, the addition of the rituximab significantly improves event-free survival among adults with CD20-positive, Philadelphia chromosome (Ph)-negative, B-cell precursor Acute Lymphoblastic Leukemia (ALL). Such advantage did not, however, translate into longer overall survival.
2. The addition of rituximab did not result in a significant increase in severe adverse events.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Though tyrosine kinase inhibitors are traditionally used to treat Ph-positive ALL, monoclonal antibodies are being increasingly used to target surface cell antigens (e.g. CD19, CD20, CD22, CD33, CD52). Rituximab is one such agent, showing significant improvement in outcomes when used to treat patients with B-cell non-Hodgkin’s Lymphoma and Burkitt’s mature B-cell lymphoma or leukemia. Though B-cells express CD20 antigen, and prior single center studies have shown benefit when adding rituximab to standard chemotherapy regimens to target such cells, no randomized prospective clinical trial has shown similar findings. As such, this study compared the trial arm of chemotherapy plus rituximab to the control arm of chemotherapy alone, for patients with Ph-negative, B-lineage ALL expressing CD20 antigen.
The trial found a lower incident of relapse in the rituximab arm. However, this did not translate into a significant difference in overall survival between the two arms. In addition, there were similar rates of adverse events between the chemotherapy plus rituximab and chemotherapy alone arms. The major strength of this study is attributed to the multi-centre, prospective randomized controlled nature of the trial, with the largest cohort of study participants to date. Limitations of this study are related to the multiple comparisons required to generate secondary endpoints and completing post-hoc analyses, and a age cohort limited to 18-59 years of age.
Click to read the study, published today in NEJM
Relevant Reading: Monoclonal antibody-based therapies: a new dawn in the treatment of acute lymphoblastic leukemia
In-Depth [randomized controlled trial]: Given prior single centre studies showing improved outcomes of adding rituximab to chemotherapy compared to chemotherapy alone, this study tested this comparison in the context of a prospective, randomized controlled trial. Adults aged 18-59 years old were accrued across 59 French and 9 Swiss sites between May 2006 and April 2014, all of which were CD20-positive, Ph-negative. Patients were randomized to receive chemotherapy either with or without rituximab, the latter of which was given through all treatment phases (total of 16-18 infusions). The primary endpoint was event-free survival. Events were defined as: failure of complete remission induction, relapse, and death.
There were a total of 209 patients enrolled in the study between the dates noted above, with 105 in the rituximab group and 104 in the chemotherapy alone group. The median percentage of CD20-positive blasts was 66% (range – 20-100%) between the two groups, and was evenly balanced. The median follow up was 30 months, with a total of 101 patients (48%) having at least one event: 44 (42%) patients in the rituximab group and 57 (55%) in the control group. Patients in the rituximab group had longer event-free compared to those in the chemotherapy alone group (HR 0.66; 95%CI 0.45-0.98; p = 0.04). The difference in survival was mostly attributed to the lower relapse rate in the rituximab group, with a subdistributon hazard ratio of 0.52 (95%CI 0.31-0.89; p = 0.02). There was no difference in overall survival between the two groups (HR 0.7; 95%CI 0.46-1.07; p = 0.1). Overall, there were 246 severe adverse events, however, there were no significant difference between the two groups (p = 0.72).
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.