Romosozumab followed by alendronate superior to alendronate alone in fracture prevention
1. In postmenopausal osteoporotic women at high risk for fracture, romosozumab followed by alendronate significantly reduced fracture risk compared to alendronate alone.
2. Although the incidence of adverse events were similar between treatment groups, serious cardiovascular adverse events were observed in more romosozumab treated patients.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Romosozumab is a bone-forming monoclonal antibody that has shown promise in early phase clinical trials for reducing the risk of fragility fractures in postmenopausal women with osteoporosis. In the Active-Controlled Fracture Study in Postmenopausal Women with Osteoporosis at High Risk (ARCH) trial, over 4000 postmenopausal women at high risk for fragility fractures were randomized to either romosozumab or alendronate arms followed by alendronate to determine the efficacy of romosozumab compared to a commonly used first-line agent. The primary endpoint, the cumulative incidence of new vertebral and clinical fractures, was significantly reduced in the romosozumab-alendronate group. This improvement in the romosozumab-alendronate group was also observed in secondary endpoints, including bone mineral density and the incidence of nonvertebral fractures. Serious cardiovascular adverse events, however, were more common in the group treated with romosozumab.
This was a phase 3, multicenter, randomized, double-blinded trial that demonstrated the potential effectiveness of romosozumab as a short-term therapy for osteoporosis. Strengths were the inclusion of osteoporotic patients with a high fracture risk and the comparison of romosozumab to an active, first-line agent. Limitations of the trial included the lack of placebo control and inability to properly evaluate cardiovascular outcomes. While fracture outcomes are improved, further work is required to better understand the cardiovascular effects of romosozumab.
Click to read the study in NEJM
Relevant Reading: Romosozumab in postmenopausal women with low bone mineral density
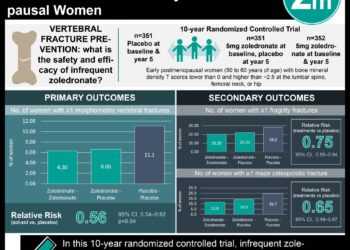
In-Depth [randomized controlled trial]: Postmenopausal women (n = 4093) with osteoporosis and a prior fragility fracture were randomly assigned to monthly subcutaneous romosozumab or weekly oral alendronate for 12 months. All patients subsequently received weekly alendronate for 12 months. The primary endpoint was the cumulative incidence of new vertebral fractures at 24 months and the cumulative incidence of clinical fractures, including nonvertebral and symptomatic vertebral fractures. Key secondary endpoints included bone mineral density at the lumbar spine, total hip, and femoral neck at 12 and 24 months and the incidence of nonvertebral fractures. Other fracture categories were evaluated as additional secondary endpoints.
Over the 24-month period, those in the romosozumab-alendronate group demonstrated a 48% lower risk of new vertebral fractures than alendronate alone (6.2% vs. 11.9%; risk ratio [RR], 0.52; 95% confidence interval [CI], 0.40 to 0.66; p < 0.001). Romosozumab-alendronate resulted in a 27% lower risk of clinical fracture than alendronate alone (hazard ratio [HR], 0.73; 95% CI, 0.61 to 0.88; p < 0.001). There was also a 19% lower risk of nonvertebral fracture than alendronate alone (HR, 0.81; 95% CI, 0.66 to 0.99; p = 0.04) and a 38% lower risk of hip fractures (HR, 0.62; 95% CI, 0.42 to 0.92; p = 0.02). Patients in the romosozumab-alendronate group demonstrated greater gains in bone mineral density from baseline that were maintained at month 36 after transitioning to alendronate (p < 0.001). The incidence of adverse events and serious adverse events were similar between the two treatment groups. However, 50 patients (2.5%) in the romosozumab group versus 38 (1.9%) in the alendronate group reported serious cardiovascular events (odds ratio [OR], 1.31; 95% CI, 0.85 to 2.00). There were no reported cases of osteonecrosis of the jaw or atypical femoral fractures during the 12-month double-blind period.
Image: CC/Wiki
©2017 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc


![Adverse pregnancy outcomes associated with thrombophilias [Classics Series]](https://www.2minutemedicine.com/wp-content/uploads/2015/07/Classics-2-Minute-Medicine-e1436017941513-75x75.png)




