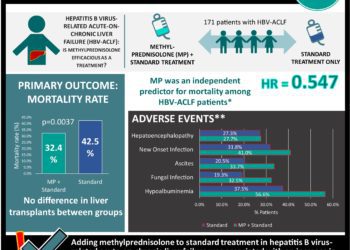
Tenofovir treatment associated with reduced perinatal transmission of hepatitis B virus
1. Perinatal transmission of hepatitis B virus (HBV) was lower in mothers treated with tenofovir disoproxil fumarate (TDF) compared to controls.
2. No significant difference in rates of birth defects found between infants of mothers receiving TDF compared to no TDF.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Serious consequences of chronic HBV infection include cirrhosis and liver cancer. Because total clearance of HBV is rare, prevention represents the best way to reduce the global burden of HBV and its long-term sequelae. In HBV positive pregnant mothers, preventing perinatal transmission with immunoprophylaxis has had a success rate between 70-90%, often linearly related to the viral load of the mother. Recent literature has provided mixed results on the use of antiviral treatment in further reducing the risk of perinatal transmission. In this randomized controlled trial, researchers investigated the use of TDF during pregnancy to prevent the perinatal transmission of HBV (of note, all infants also received immunoprophylaxis).
Their results demonstrated a significantly lower rate of HBV transmission in the TDF group compared to the control group, and no differences in the rates of birth defects. Additionally, researchers found that mothers in the HBV group had significantly lower levels of HBV DNA in the TDF group at 28 weeks postpartum compared to the control. However, rates of seroconversion or loss of Hepatitis B envelop antigen (HBeAg) and Hepatitis B surface antigen (HBsAg) did not differ significantly between the two groups. Overall, this study demonstrated that treatment with tenofovir during late pregnancy in HBV positive mothers significantly reduced perinatal transmission without increased rates of birth defects.
Click to read the study, published today in NEJM
Relevant Reading: Telbivudine or lamivudine use in late pregnancy safely reduces perinatal transmission of hepatitis B virus in real-life practice
In-Depth [randomized controlled trial]: In this study, researchers recruited 200 HBV positive mothers, who were randomly assigned to either treatment with TDF or standard prenatal care without TDF between 2012-2013. Primary outcomes included rates of perinatal transmission at 28 weeks postpartum and birth defects. In the intention-to-treat analysis, the rate of HBV transmission of 5% (5 of 97) in TDF group infants was significantly lower than the 18% transmission rate of 18% (18 of 100; 95%CI 1-10; p = 0.01) in the control group. For birth defects, 2% (2 of 95) of the infants in the TDF group versus 1% (1 of 88) of infants in the control group had birth defects (p = 0.01). Secondary outcomes included the percentage of mothers with an HBV DNA level ≤200,000 IU/mL at delivery and the rate of HbeAg or HBsAg loss or seroconversion at 28 weeks postpartum. 68% (66 of 97) of mothers in the TDF group versus 2% (2 of 100) of the control group had HBV DNA levels ≤200,000 IU/mL. The rates of serocoversion or loss of HBeAg or HBsAg status were not significantly different between the two groups. Maternal adverse events included significantly higher rates of creatinine kinase in the TDF (7%) compared to the control group (0%; p = 0.006), and significantly higher rates of alaninetransferase levels in the TDF group (45%) compared to the control group (30%; p = 0.03).
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.