The DAPA-HF trial: Dapagliflozin in patients with heart failure [Classics Series]
This study summary is an excerpt from the book 2 Minute Medicine’s The Classics in Medicine: Summaries of the Landmark Trials
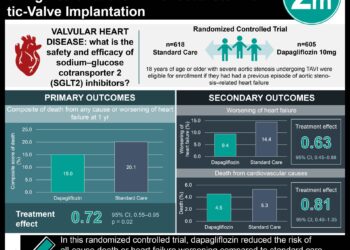
1. This study found that patients with heart failure with reduced ejection fraction (HFrEF) who received dapagliflozin had lower rates of composite cardiovascular death and worsening heart failure (16.3%) compared to controls (21.1%).
2. Presence or absence of diabetes had no significant impact on the outcomes in either group.
Original Date of Publication: November 2019
Study Rundown: The Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure (DAPA-HF) trial evaluated the cardioprotective effects of the sodium–glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin in patents with HFrEF. Composite cardiovascular death and worsening heart failure was significantly lower in those taking dapagliflozin as compared to placebo. Although SGLT2 inhibitors are typically used in diabetic patients, the presence or absence of diabetes did not alter outcomes in either group. The secondary composite outcome of cardiovascular death and hospitalizations for heart failure was also significantly reduced in the dapagliflozin group. The main limitation of the DAPA-HF trail was limited diversity in the study population, possibly impacting generalizability of the results. The DAPA-HF trial was the first study to demonstrate that in HFrEF patients both with and without diabetes, dapagliflozin reduces progression of heart failure, associated hospitalizations, and risk of cardiovascular death.
Click to read the study in NEJM
In-Depth [randomized control trial]: The DAPA-HF trial was conducted on a group of HFrEF patients (n = 4744) who were randomized into either a dapagliflozin group (n = 2373) or placebo (n = 2371) for a median duration of follow-up of 18.2 months. All trial outcomes were adjudicated by a blinded clinical-events committee. The primary composite endpoint of cardiovascular death and worsening heart failure characterized by hospitalization or urgent primary care visit resulting in intravenous therapy was significantly lower in the dapagliflozin group (16.3%) than the placebo group (21.2%) (HR 0.74; 95%CI 0.65-0.85). The dapagliflozin group also had a significantly reduced rate of the composite outcome of cardiovascular death and hospitalization due to heart failure (HR 0.75; 95%CI 0.65-0.85). Both groups were matched at screening for type 2 diabetes (42%) and diabetes status did not have any impact on the primary or the secondary composite outcomes.
McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. New England Journal of Medicine. 2019 Nov 21;381(21):1995–2008.
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.