USPSTF recommends aspirin for women at risk for preeclampsia
1. Use of low-dose aspirin was associated with a reduced risk for preeclampsia, intrauterine growth restriction and preterm birth in women at high risk for preeclampsia.
2. Low-dose aspirin was not associated with any appreciable harms during pregnancy.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Preeclampsia is among the leading causes of maternal and perinatal morbidity and mortality worldwide. It accounts for 15% of preterm births in the United States, and is more common in women with a history of preeclampsia, comorbid conditions (e.g. chronic hypertension), or multifetal gestation. A meta-analysis of several randomized control trials (RCTs) conducted by the U.S. Preventive Services Task Force (USPSTF) suggested that initiation of low-dose aspirin therapy in women at high risk for preeclampsia reduced the risk of preeclampsia by 24%, preterm birth by 14%, and intrauterine growth restriction (IUGR) by 20%. The agency also found that using low-dose aspirin during pregnancy is not associated with increased risk of placental abruption, postpartum hemorrhage, fetal intracranial bleeding or perinatal mortality. Based on these findings, the USPSTF recommends the daily use of low-dose aspirin during pregnancy to reduce the risk of preeclampsia. This recommendation is limited by the lack of a defined, validated metric for determining a woman’s risk for preeclampsia. It is also weakened by the fact that a majority of the studies examined did not include a racially diverse sample population, especially regarding non-Hispanic black women, who are at a higher risk for preeclampsia than white women.
Click to read the study, published today in the Annals of Internal Medicine
Relevant Reading: Early administration of low-dose aspirin for the prevention of severe and mild preeclampsia: a systematic review and meta-analysis
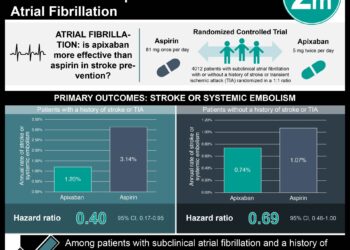
In-Depth [systematic review]: The USPSTF reviewed the data from fifteen RCTs to determine whether low-dose aspirin use had any positive or negative effects on the incidence of preeclampsia. Pooled analyses of the studies’ findings showed that low-dose aspirin was associated with a 24% reduction in risk for preeclampsia (relative risk (RR) 0.76), a 20% reduction in risk for IUGR (RR 0.80) and a 14% reduction in risk for preterm birth (RR 0.86) in women at increased risk for preeclampsia. The USPSTF also reviewed nineteen RCTs to evaluate short-term harms of low-dose aspirin use during pregnancy. Pooled analyses suggested no statistically significant increases in placental abruption (RR, 1.17), perinatal mortality (RR, 0.92) or postpartum hemorrhage (RR, 1.02) among women taking low-dose aspirin during pregnancy. There was insufficient evidence identified on long-term developmental outcomes in offspring exposed to aspirin in utero.
More from this author: Marked decline in cystic fibrosis-related mortality over 10 years, ACP endorses polysomnography over portable monitors for diagnosing sleep apnea, New ACP guidelines recommend against regular pelvic exams, Insufficient evidence for dementia screening tests in older adults, Insufficient evidence for vitamin supplements in prevention of CVD or cancer
Image: PD
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.