Viral vector based gene therapies show promise for treating focal epilepsy [PreClinical]
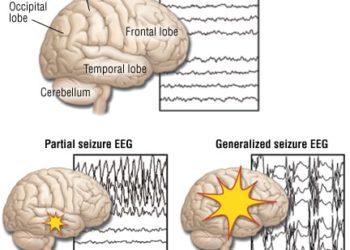
1. Seizures are often associated with accumulation of extracellular glutamate due to the synchronous discharges of excitatory neurons.
2. Treatment of rats with a lentiviral plasmid encoding a glutamate-gated chloride channel was able to decrease focal seizure activity by hyperpolarizing neurons and decreasing their effective membrane resistance.
Evidence Rating Level: 2 (Good)
Study Rundown: Despite the development of several anti-epileptic medications, one-third of patients with epilepsy continue to have seizures. Current antiepileptic drugs have a narrow therapeutic window and require close monitoring. Surgical resection of epileptogenic foci is restricted to cases in which the foci are far enough away from areas involved in motor or sensory processing. For patients with treatment-refractory focal seizures, molecular strategies aimed at decreasing the effect of glutamate accumulation near epileptogenic foci in the brain, may provide a new and potentially more effective approach at reducing seizure activity.
In this study, a plasmid was designed to carry the sequence of a glutamate-gated chloride channel (GluCl) from C. elegans. A residue in the α subunit of the channel was mutated to enhance the channel’s glutamate sensitivity (eGluCl). Expression of eGluCl was placed under the control of the human CaMKIIα promoter in order to bias expression of eGluCl to excitatory neurons. The plasmid was delivered to neurons via lentivirus, and the eGluCl channel was found to be stably expressed mainly at non-synaptic sites on neuronal cell surfaces.
The ability of the eGluCl treatment to attenuate seizure development was assessed in rodent models, in which seizures were induced in two ways: (1) with intracortical injection of the chemoconvulsant pilocarpine, which acutely induces seizure activity or (2) with tetanus-toxin injection into the visual cortex which results in chronic spontaneous seizures over the course of several weeks. Administration of eGluCl was found to decrease acute chemoconvulsant-induced seizures, as well as decrease the absolute number of seizures in the model of chronic focal neocortical epilepsy. Additionally, the eGluCl treatment itself was found to not have a major effect on normal brain behavioral function, as assessed by two tests of motor coordination, suggesting that gene therapy with eGluCl may be well-tolerated. While further studies in higher-order primates will be necessary to better predict its effectiveness and off-target effects in humans, the ability of eGluCl therapy to selectively inhibit neurons in response to increases in extracellular glutamate concentrations represents a promising new approach to focal epilepsy that remains refractory to other forms of treatment.
Click here to read about the eGluCl therapy for focal epilepsy
Relevant Reading: Novel approaches to epilepsy treatment
In-Depth [animal study]: A wild-type glutamate-gated chloride channel from C. elegans GluCl was codon optimized for human expression (GluCl) and the L9’F mutation to increase glutamate sensitivity (eGluCl) was inserted with a QuikChange II kit from Agilent. The increased sensitivity of the eGluCl under the control of the human CamKII-α promoter was confirmed with voltage clamp recordings using a mouse neuroblastoma cell line (Neuro-2a).
In order to induce seizure activity, male Sprague Dawley rats were cannulated either in the forelimb area of the right primary motor cortex or in the right visual cortex, and an ECoG transmitter was implanted to allow for wireless telemetry readings. For the chemoconvulsant seizure model, animals were injected on four consecutive days with increasing amounts of pilocarpine solution into the primary motor cortex. Three days later, all animals were injected with either eGluCl lentivector (n = 10) or control GFP lentivector (n = 10) at the same location. Two weeks later, the chemoconvulsant injection procedure was repeated and compared to pre-treatment seizure activity. This experimental design allowed normalization of every pilocarpine-invoked seizure to its corresponding pre-treatment seizure. Animals treated with eGluCl were found to exhibit a decrease in the electrocorticogram (ECoG) coastline, an aggregate of frequency and amplitude, between the two pilocarpine injections and displayed a decrease in spike frequency (p = 0.007).
For the chronic tetanus toxin epilepsy model, animals were injected with tetanus toxin in the right visual cortex. Eleven days after tetanus toxin injection, animals were injected with eGluCl lentivector or GFP lentivector. eGluCl significantly decreased the number of subsequent seizures compared to GFP-treated animals (P < 0.001), though there was no significant difference in average seizure duration or severity between the two groups.
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.




![Tumor-specific mutant antigens are potential targets for cancer therapy [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2014/12/mouse-e1417555547919-75x75.jpg)