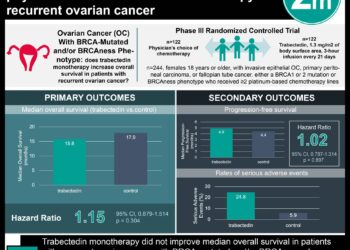
#VisualAbstract: Adavosertib plus gemcitabine improves survival in platinum-resistant recurrent ovarian cancer
1. Adavosertib plus gemcitabine showed improved progression-free and overall survival in platinum-resistant or refractory recurrent ovarian cancer versus gemcitabine plus placebo.
2. Adavosertib plus gemcitabine was associated with more hematological adverse effects, such as neutropenia, versus control.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Despite advances in the field of oncology, ovarian cancer is amongst the leading causes of death from gynecological malignancies around the world. While platinum-based chemotherapy is generally considered the standard of care, recurrence and subsequent treatment resistance is common. A new approach has been to target molecules involved in the DNA damage response to overcome the resistance to platinum therapy. Both adavosertib and gemcitabine are two such molecules that have shown synergistic effects in preclinical studies of ovarian cancer. This phase 2, double-blind, randomized, controlled trial compared the use of adavosertib plus gemcitabine versus gemcitabine plus placebo in patients with platinum-resistant or refractory recurrent ovarian cancer. Overall, participants in the adavosertib plus gemcitabine group showed improvements in median progression-free and overall survival versus control. The former also showed an improved response according to the cancer antigen-125 (CA-125) criteria. In general, the adavosertib group had more adverse events than the control group; the most common grade 3 or greater adverse events included hematological events and fatigue. Grade 3-4 neutropenias and thrombocytopenias were also more common in the adavosertib group. No treatment-related deaths were recorded. A post-hoc analysis also demonstrated that patients with homologous recombination gene mutations were more likely to respond to the adavosertib plus gemcitabine combination, resulting in a nonsignificant increase in progression-free survival. A notable limitation to this study was the relatively small sample size. Because of this, the researchers were unable to implement stratification factors in their study design. This is an important concern given that, for example, BRCA mutation status is a known prognostic factor not considered in this study.
Click to read the study in The Lancet
Click to read an accompanying editorial in The Lancet
Relevant Reading: Wee1 is a novel independent prognostic marker of poor survival in post-chemotherapy ovarian carcinoma effusions.
In-Depth [randomized controlled trial]: This double-blind, randomized, placebo-controlled phase 2 trial took place in 11 academic centers in the USA and Canada, and recruited 124 women with platinum-resistant or refractory recurrent ovarian cancer. While 99 participants with high-grade serous ovarian cancer were randomized to the adavosertib plus gemcitabine or the gemcitabine plus placebo group, the other 25 participants with non-high-grade serous ovarian cancer were placed in an exploratory cohort with adavosertib and gemcitabine. The primary endpoint of this study was progression-free survival, with secondary outcomes being response rate, overall survival at 1 year, and safety/tolerability.
Overall, median progression-free survival was improved in the adavosertib group (4.6 months [95% confidence interval [CI] 3.6-6.4] vs. 3.0 months [95% CI 1.8-3.8], hazard ratio = 0.55 [95% CI 0.35-0.90]; p=0.015). Median overall survival was also improved in the adavosertib group (11.4 months [95% CI 8.2-16.5] vs. 7.2 months [95% CI 5.2-13.2], hazard ratio = 0.56 [95% CI=0.35-0.91]; p=0.017). A nonsignificant improved response by CA-125 criteria was seen in the adavosertib group (23% vs. 9%, p=0.16). The participants in the gemcitabine plus adavosertib group had more grade 3 or higher adverse events than gemcitabine with placebo. Examples include grade 3-4 neutropenia (62% with adavosertib vs. 30% with control) and thrombocytopenia (31% with adavosertib vs. 6% with control). In a post-hoc analysis, patients with homologous recombination gene mutations, such as CCNE1, had an improved response to adavosertib (Fisher’s exact test p =0.013). This was also associated with a nonsignificant increase in progression-free survival (log-rank p=0.13).
©2020 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.