#VisualAbstract: Longer-Term Assessment of Azithromycin for Reducing Childhood Mortality in Africa
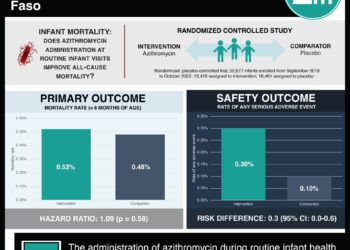
1. Mass, twice-yearly administration of azithromycin to children between 1 and 59 months of age in communities in Niger previously not administered azithromycin significantly reduced childhood mortality rates.
2. Communities given azithromycin for a third year, after receiving it the previous 2 years, had no significant change in childhood mortality rates.
Evidence Rating Level: 1 (Excellent)
Study Rundown: A prior, related tria lMORDOR I showed that twice-yearly azithromycin administration to children in various African countries reduced childhood mortality significantly. There was possibility that if administration were continued, antibiotic resistance could develop and reduce the efficacy of treatment. There was also the potential for increased efficacy through reduction of sensitive bacteria in the community. The MORDOR II (Macrolides Oraux pour Réduire les Décès avec un Oeil sur la Résistance) trial evaluated childhood mortality in various Niger communities who had and had not received azithromycin the previous 2 years. Childhood mortality rates in communities who had and had not previously received azithromycin were similar. In communities not previously administered azithromycin notable reductions in childhood mortality rates were observed.
This study provides additional follow-up to suggest continual community antibiotic treatment in certain countries may reduce childhood mortality rates. It is limited in scope of study to community level data and by a single country sample as opposed to multiple countries in the previous associated trial. Long term effects will also need to be evaluated prior to considering wider administration schemes.
Click to read the study in NEJM
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.