#VisualAbstract: Sonelokimab showed significant clinical benefit over placebo in patients with plaque psoriasis
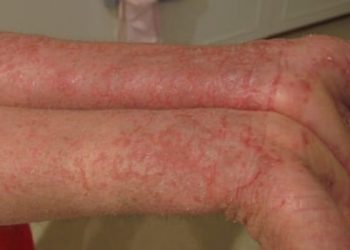
1. At 12 weeks, the proportion of patients with an IGA score of 0 or 1 was significantly greater in the sonelokimab groups than in the placebo group.
2. No dose-response associations with toxicity were apparent for the sonelokimab groups.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Sonelokimab is a novel trivalent nanobody comprised of monovalent moieties specific to human interleukin (IL)-17A, IL-17F, and human serum albumin. Studies assessing the inhibition of binding of IL-17A to IL-17RA have shown increased efficacy with sonelokimab, allowing it to be used as a potential treatment for plaque-type psoriasis. This randomized controlled trial aimed to assess the safety, efficacy, and tolerability of sonelokimab versus placebo in patients with plaque-type psoriasis, with secukinumab serving as an active control. The primary outcome was the proportion of patients receiving sonelokimab with an Investigator’s Global Assessment (IGA) score of 0 or 1 at week 12, while secondary outcomes included Psoriasis Area & Severity Index (PASI) responses, change in body surface area of psoriasis, and safety evaluations. According to study results, sonelokimab 120 mg or less showed significant clinical benefit over placebo, with respect to time-to-onset of treatment, durable improvements, and acceptable safety profile. A limitation of this study is that it did not assess the effect of either treatment on systemic diseases of plaque psoriasis such as metabolic syndrome and cardiovascular disease. Nevertheless, this study expands our understanding of the use of trivalent nanobodies for the treatment of plaque psoriasis.
Click to read the study in The Lancet
Relevant Reading: Bimekizumab versus Secukinumab in Plaque Psoriasis
In-depth [randomized controlled trial]: Between Aug 15, 2018, and Mar 27, 2019, 383 patients were assessed across 41 clinics in seven countries. Included patients were those aged 18-75 years with stable moderate to severe plaque-type psoriasis (defined as IGA ≥ 3, body surface area involvement ≥ 10%, and PASI ≥ 12) for ≥ 6 months before randomization. Those previously treated with ≥ 2 biologics or therapy targeting IL-17 were excluded. Altogether, 313 patients (52 in the placebo, sonelokimab 30 mg and 60 mg groups, 53 in the sonelokimab 120 mg normal load and secukinumab 300 mg groups, and 51 in the sonelokimab 120 mg augmented load group) were included in the analysis. Baseline characteristics were similar across groups.
At week 12, 0.0% (95% confidence interval [CI] 0.0-6.8, p<0.0001) patients in the placebo group had an IGA score of 0 or 1 compared to 48.1% (95% CI 34.0-62.4, p<0.0001) in the sonelokimab 30 mg group, 84.6% (95% CI 71.9-93.1, p<0.0001) in the sonelokimab 60 mg group, 77.4% (95% CI 63.8-87.7, p<0.0001) in the sonelokimab 120 mg normal load group, 88.2% (95% CI 76.1-95.6, p<0.0001) in the sonelokimab 120 mg augmented load group, and 77.4% (95% CI 63.8-87.7, p<0.0001) in the secukinumab 300 mg group. During the induction period, 49.5% (155 of 313) patients had one or more mild to moderate adverse events; the most frequent being nasopharyngitis (13.5%), pruritis (6.7%), and upper respiratory tract infection (4.3%) in all patients on sonelokimab during weeks 0-12. Over the 52-week study period, sonelokimab safety was similar to secukinumab, with the only exception being manageable Candida infections (1.9% in the secukinumab group vs. 17.4% in all sonelokimab-containing groups). Findings from this study suggest that sonelokimab may be superior to placebo for treatment of plaque-related psoriasis.
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.