Weekly dalbavancin noninferior to daily therapy for acute skin infection [DISCOVER 1 and 2 Trials]
1. Once-weekly dalbavancin was not inferior to twice-daily vancomycin and linezolid in treating acute bacterial skin infection.
2. Fewer adverse events were reported in patients treated with dalbavancin compared to those treated with vancomycin and linezolid.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Cellulitis, an infection of the skin and soft tissues, is a common reason for hospitalization. It is most often caused by gram-positive bacteria that normally reside on skin surfaces and mucous membranes. These pathogens can cause infection if they penetrate external barriers through injuries or lesions and can be difficult to treat in severe cases. A particular challenge is the treatment of skin infection with methicillin-resistant Staphylococcus aureus. Dalbavancin is a lipoglycopeptide antibiotic with activity against gram-positive pathogens. Dalbavancin has a long plasma half-life allowing for less frequent dosing compared to the current standard of daily therapy. DISCOVER 1 and DISCOVER 2 are non-inferiority trials with identical designs, aiming to demonstrate the efficacy of once-weekly intravenous dalbavancin compared to daily conventional therapy. The pooled analysis of these two trials found that once-weekly dalbavancin was not inferior to a twice-daily protocol of vancomycin and linezolid in treating acute bacterial skin infection and infection of skin structures. The identical design of these two trials allowed the investigators to pool data for better estimates of efficacy and rates of adverse events. The trials used a newly derived and more objective primary end point assessing lesion size and absence of fever as opposed to investigator-assessed response to treatment which has been used in the past.
Click to read the study, published today in NEJM
Click to read an accompanying editorial in NEJM
In-Depth [randomized controlled trial]: Collectively, the two trials included 1312 patients who were randomized to receive dalbavancin intravenously on days 1 and 8, or vancomycin intravenously for 3 days followed by an option to switch to oral linezolid to up to 14 days of therapy. Patients were eligible if they had a diagnosis of an acute bacterial skin or skin-structure infection, would require at least 3 days of intravenous therapy, and had one or more systemic signs of infection. The criteria for the primary end point of early clinical response to therapy were the cessation of spread of infection-related erythema and absence of fever at 48 to 72 hours. In the pooled analysis, 79.7% of patients in the dalbavancin group and 79.8% of patients in the vancomycin-linezolid group demonstrated an early clinical response (weighted difference, -0.1 percentage points; 95% CI, -4.5 to 4.2). Outcomes in each trial individually were similar to results from the pooled analysis. Fewer adverse events were reported in the dalbavancin group than in the vancomycin-linezolid group; however, rates of discontinuation due to an adverse event were similar among the groups (2.1% in the dalbavancin group and 2.0% in the vancomycin-linezolid group).
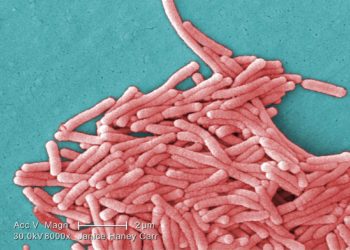
Image: PD/CDC. MRSA
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.